Cleaning and Disinfection Revalidation Triggers
1. Purpose
Revalidation triggers define the conditions under which cleaning and disinfection processes must be reassessed and revalidated to ensure continued effectiveness.
The objective is to maintain the validated state by ensuring that any change or emerging risk does not compromise:
- residue removal capability
- disinfectant effectiveness
- overall contamination control
2. Role in Lifecycle Control
Revalidation is not routine. It is event-driven and based on:
- changes that may impact performance
- evidence of loss of control
- periodic reassessment outcomes
Triggers ensure that validation remains aligned with actual operating conditions.
3. Categories of Revalidation Triggers
3.1 Product and Process Changes
Changes in products or manufacturing conditions may affect cleanability.
Examples:
- introduction of new products
- changes in formulation
- increased batch size
- changes in process parameters
These changes may alter residue characteristics or cleaning difficulty.
3.2 Equipment and Facility Changes
Modifications to equipment or surfaces can impact cleaning and disinfection effectiveness.
Examples:
- new equipment or replacement
- modification of product-contact surfaces
- changes in surface materials or finishes
- facility modifications affecting cleaning or disinfection practices
3.3 Cleaning Process Changes
Changes to cleaning procedures must be evaluated.
Examples:
- change in cleaning agents or disinfectants
- modification of concentrations
- changes in cleaning methods or equipment
- changes in contact time or temperature
Any change that may impact performance requires reassessment.
3.4 Disinfection Program Changes
Specific to microbiological control:
- introduction of new disinfectants
- changes in disinfectant rotation
- modification of application methods
- changes in contact time
These changes require confirmation of continued efficacy.
3.5 Analytical and Sampling Changes
Changes in analytical or sampling methods may affect data reliability.
Examples:
- new analytical methods
- changes in detection limits or LOQ
- modification of sampling techniques
- changes in recovery assumptions
Revalidation ensures comparability and validity of results.
3.6 Deviations and Failures
Evidence of inadequate performance is a direct trigger.
Examples:
- cleaning failures or out-of-limit results
- repeated deviations
- investigation findings indicating process weakness
These events require reassessment and potential revalidation.
3.7 Environmental Monitoring Trends
For disinfection control:
- increasing microbial counts
- shifts in microbial population
- detection of resistant organisms
These trends may indicate reduced disinfectant effectiveness.
3.8 Periodic Review Outcomes
Periodic reviews may identify the need for revalidation.
Examples:
- negative trends in cleaning verification data
- accumulation of minor deviations
- emerging risks not previously considered
4. Risk-Based Evaluation
Not all triggers require full revalidation.
Each trigger must be assessed based on:
- impact on cleaning or disinfection effectiveness
- extent of change
- existing validation data
- risk to product quality
Possible outcomes:
- no action required
- limited verification
- partial revalidation
- full revalidation
Revalidation triggers must be evaluated through a structured decision process to determine the appropriate level of response.
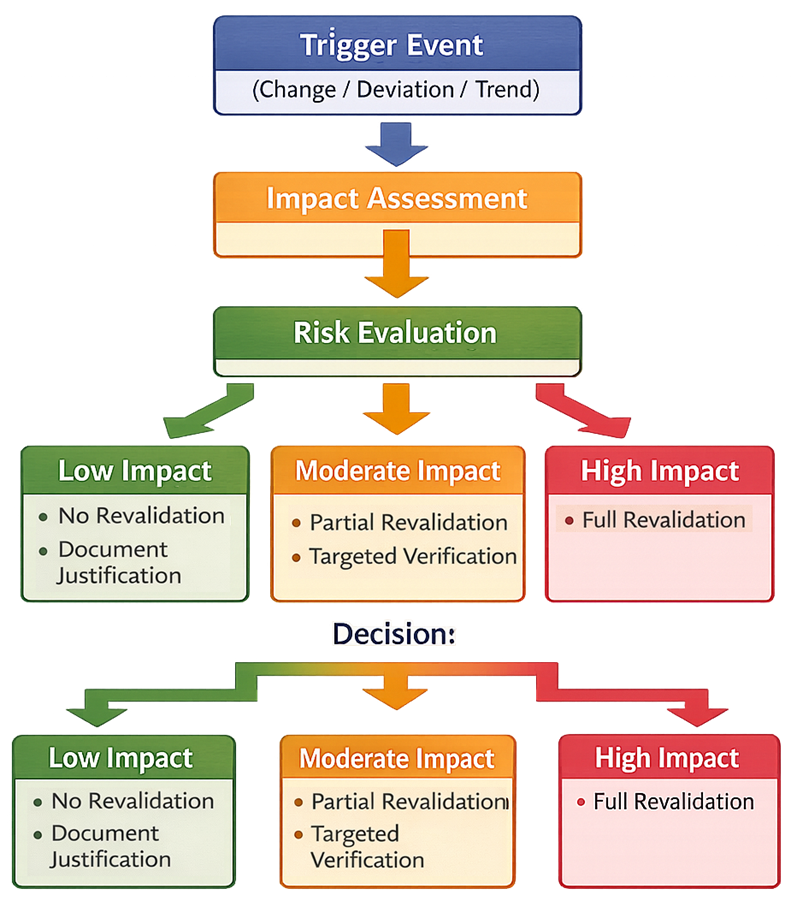
Trigger events are assessed for their impact on cleaning and disinfection effectiveness. Based on risk, actions may range from no revalidation to partial or full revalidation. This ensures that revalidation efforts are proportional and justified
5. Scope of Revalidation
Revalidation scope must be defined based on risk.
Options include:
- targeted evaluation of specific equipment or products
- repeat of cleaning efficacy studies
- repeat of disinfectant efficacy studies
- reassessment of sampling and analytical methods
The scope must be justified and documented.
6. Documentation Requirements
All revalidation triggers must be documented.
Required elements include:
- description of the change or event
- impact assessment
- justification of revalidation decision
- defined scope of revalidation
- results and conclusions
Traceability must link triggers to actions taken.
7. Common Deficiencies
Typical issues include:
- failure to define clear triggers
- inconsistent application of revalidation decisions
- lack of documented rationale
- over-reliance on periodic review without action
- failure to reassess after significant changes
These deficiencies lead to loss of validated state.
8. Key Principle
Revalidation triggers ensure that validation remains dynamic and responsive to change.
They provide a controlled mechanism to reassess processes when risk or conditions evolve.

