Detection Limits
1. Purpose and Definition
Detection limits define the lowest amount of residue that an analytical method can reliably detect or quantify. In cleaning validation, they ensure that analytical methods are capable of measuring residues at or below established acceptance criteria.
Detection capability must be aligned with health-based limits to ensure that residues are not only controlled but also measurable.
2. Key Terminology
Two primary limits are used:
- Limit of Detection (LOD)
the lowest amount of analyte that can be detected but not necessarily quantified - Limit of Quantitation (LOQ)
the lowest amount of analyte that can be quantitatively measured with acceptable accuracy and precision
LOQ is the critical parameter for cleaning validation because results must be numerically reported and compared to limits.
Detection and quantitation limits are distinguished by the ability to reliably measure the analytical signal above background noise.
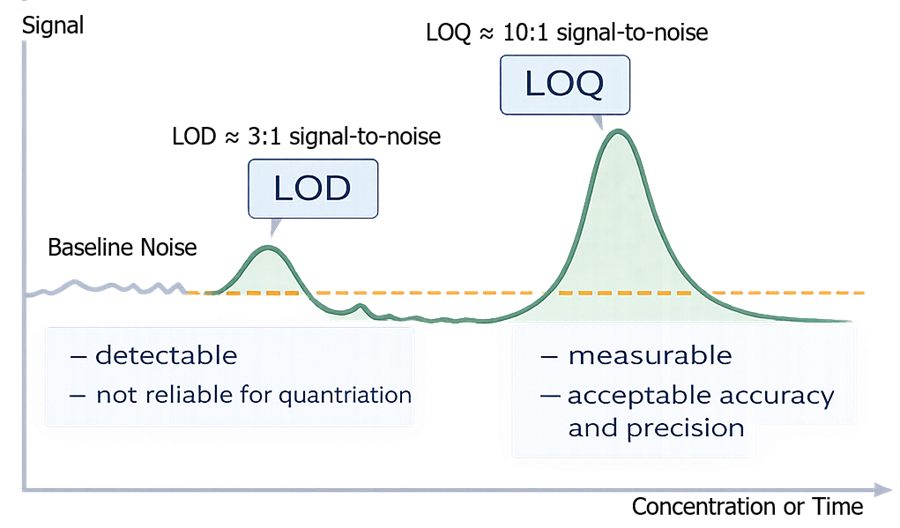
LOD represents the point at which a signal can be distinguished from baseline noise, while LOQ represents the point at which the signal can be measured with acceptable accuracy and precision. The distinction is critical for interpreting analytical results.
3. Role in Cleaning Validation
Detection limits ensure that:
- analytical methods can detect residues below acceptance criteria
- absence of detected residue is meaningful
- results are suitable for decision-making
If the method cannot detect residues at required levels, validation results are not reliable.
4. Relationship to Acceptance Criteria
Detection limits must be significantly lower than the established residue limits.
Typical expectation:
- LOQ ≤ 50% of acceptance limit
- preferably lower for high-risk products
This ensures adequate sensitivity and confidence in results.
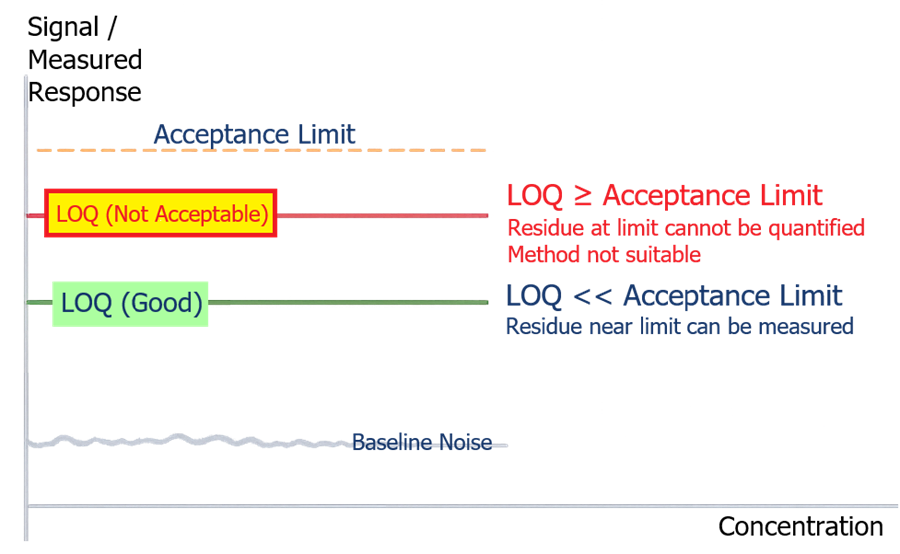
The limit of quantitation must be lower than the acceptance criterion to ensure that residues approaching the limit can be reliably measured. If LOQ is equal to or higher than the acceptance limit, the method cannot confirm compliance.
5. Determination of LOD and LOQ
Detection limits are determined experimentally using validated analytical methods.
Common approaches include:
- signal-to-noise ratio method
- LOD ≈ 3:1
- LOQ ≈ 10:1
- standard deviation of response and slope method
- calibration curve analysis
The selected approach must be justified and documented.
6. Matrix and Sampling Effects
Detection limits must consider the full sample matrix:
- swab extraction solutions
- rinse solvents
- potential interferences from cleaning agents
Matrix effects may increase background noise and reduce sensitivity.
Detection limits determined in pure solutions are not sufficient.
7. Method Capability
Detection capability depends on:
- analytical technique such as HPLC, TOC, UV
- instrument sensitivity
- sample preparation and extraction efficiency
- operator and system variability
The entire method, not just the instrument, defines detection limits.
8. Practical Application
Detection limits are applied to:
- verify method suitability before validation
- confirm that sampling results are meaningful
- support interpretation of non-detect results
A “non-detect” result is only valid if the method can detect below the required limit.
9. Common Deficiencies
Typical issues include:
- LOQ higher than acceptance criteria
- failure to evaluate matrix effects
- reliance on theoretical rather than experimental values
- inadequate documentation of determination method
These deficiencies invalidate analytical results.
10. Documentation Requirements
Detection limits must be fully documented.
Required elements include:
- method used for determination
- experimental data and calculations
- matrix conditions
- justification of selected limits
Traceability must support verification and review.
11. Lifecycle Considerations
Detection limits must be reassessed when:
- analytical methods change
- instruments are replaced or modified
- new products or matrices are introduced
Method capability must remain aligned with cleaning validation requirements.
12. Key Principle
Detection limits define what can be seen and measured.
If residues cannot be detected at required levels, cleaning validation cannot be demonstrated.

