Disinfectant Efficacy Studies
1. Purpose and Definition
Disinfectant efficacy studies demonstrate that selected disinfectants are capable of reducing or eliminating microorganisms on cleanroom surfaces under defined conditions. These studies confirm that disinfection procedures provide effective microbiological control and support contamination control strategies.
Effectiveness is expressed as log reduction of viable microorganisms following exposure to the disinfectant.
2. Role in Contamination Control
Disinfectant efficacy studies are a key component of cleanroom control. They:
- verify effectiveness of disinfectants used in the facility
- support selection and rotation of disinfectants
- confirm required contact times
- demonstrate control of environmental microorganisms
They provide direct evidence that disinfection practices are capable of maintaining microbiological control.
3. Study Design — Surface Coupon Approach
Efficacy is typically evaluated using surface coupon studies.
Coupons represent materials used in the cleanroom, such as:
- stainless steel
- glass
- polymers and elastomers
- epoxy or coated surfaces
Coupons must be representative of actual equipment and facility surfaces.
4. Selection of Microorganisms
Studies must include microorganisms relevant to the process. Categories include:
- standard challenge organisms (ATCC strains)
well-characterized reference strains obtained from American Type Culture Collection, representing a range of resistance profiles and commonly used to demonstrate broad-spectrum disinfectant effectiveness - facility isolates
microorganisms recovered from the cleanroom environment, representing actual site-specific contamination risks - molds and yeasts where applicable, particularly for areas with elevated risk of fungal contamination
Use of ATCC strains ensures reproducibility and comparability of results, while inclusion of facility isolates ensures that disinfectants are effective against microorganisms actually present in the facility.
5. Inoculation and Preparation
Coupons are inoculated with a known population of microorganisms.
Key steps:
- apply defined inoculum to coupon surface
- allow drying under controlled conditions
- ensure uniform distribution of microorganisms
Drying simulates real contamination conditions and increases challenge severity.
6. Disinfectant Application
Disinfectant is applied under defined conditions:
- concentration as used in practice
- application method consistent with SOPs
- defined contact time
Multiple exposure times may be evaluated to determine minimum effective contact time.
7. Neutralization and Recovery
After exposure:
- disinfectant activity must be neutralized
- surviving microorganisms must be recovered from the surface
Neutralization is critical to prevent continued antimicrobial activity during analysis. Recovery methods must be validated to ensure accurate enumeration.
Disinfectant efficacy studies are performed using a structured coupon-based workflow that simulates surface contamination and evaluates microbial reduction.
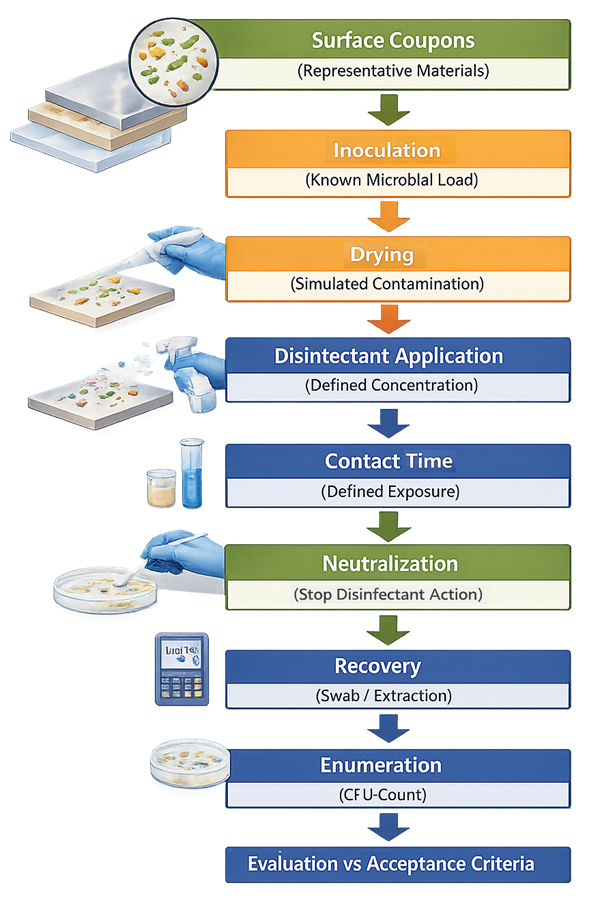
Representative surface coupons are inoculated with a known microbial load and allowed to dry. Disinfectant is applied under defined conditions and exposure times, followed by neutralization to stop antimicrobial activity. Surviving microorganisms are recovered and enumerated, and log reduction is calculated to determine disinfectant effectiveness.
8. Enumeration and Log Reduction
Recovered microorganisms are quantified and compared to initial inoculum.
Log reduction is calculated as:
- reduction in viable count relative to initial population
Typical expectations:
- defined log reduction based on application and regulatory expectations
- consistent performance across replicates
9. Acceptance Criteria
Acceptance criteria must be predefined. They typically include:
- minimum log reduction for bacteria
- minimum log reduction for fungi and molds
- effectiveness across all tested surfaces
Criteria must reflect intended use and risk level of the cleanroom.
10. Factors Affecting Efficacy
Disinfectant performance depends on:
- surface material and roughness
- organic residue or soil
- microorganism type and resistance
- disinfectant concentration
- contact time
These factors must be considered in study design.
11. Disinfectant Rotation
Studies may support rotation programs.
- different disinfectants target different organisms
- rotation reduces risk of resistance
- efficacy must be demonstrated for each disinfectant
12. Documentation Requirements
Studies must be fully documented.
Required elements include:
- study protocol and design
- coupon materials and preparation
- microorganisms used
- inoculation levels
- disinfectant conditions
- recovery and enumeration data
- calculated log reductions
Documentation must support traceability and review.
13. Common Deficiencies
Typical issues include:
- lack of facility isolates
- inadequate neutralization validation
- unrealistic contact times
- poor recovery efficiency
- insufficient replication
These deficiencies reduce confidence in results.
14. Lifecycle Considerations
Disinfectant efficacy must be reassessed when:
- new disinfectants are introduced
- formulations or concentrations change
- new materials are introduced into the cleanroom
- environmental monitoring trends change
Studies must remain representative of current conditions.
15. Key Principle
Disinfectant efficacy studies demonstrate microbial kill under defined conditions.
They provide direct evidence that disinfection practices are effective and capable of controlling contamination in the cleanroom environment.

