Drying Equipment
Drying is a critical unit operation in pharmaceutical solid dosage manufacturing. Following wet granulation, granules contain residual solvent or water from the binder solution. The drying process removes this moisture to achieve a stable granule structure with controlled residual moisture content suitable for downstream processing such as milling, blending, and compression.
Proper drying is essential because residual moisture directly affects granule strength, flow properties, compressibility, and chemical stability of the active pharmaceutical ingredient. Excess moisture may lead to granule agglomeration, poor flow, and tablet defects, while excessive drying can produce brittle granules and excessive fines. Drying equipment must therefore provide controlled heat and mass transfer conditions that allow reproducible moisture removal while maintaining product quality.
Drying systems used in pharmaceutical manufacturing must also support controlled airflow, uniform temperature distribution, containment of product particles, and hygienic equipment design consistent with cGMP requirements.
1. Purpose of Drying in Solid Dosage Manufacturing
The primary objective of the drying step is to reduce moisture content in wet granules to a defined level that ensures stable material handling and product quality. Drying serves several important functions:
- Removal of solvent or water introduced during wet granulation
- Stabilization of granule structure and mechanical strength
- Improvement of granule flowability
- Prevention of microbial growth and chemical degradation
- Preparation of granules for downstream milling and blending operations
Moisture content must be controlled within defined limits because excessive residual moisture can lead to sticking during tablet compression, while insufficient moisture can reduce granule cohesion and produce excessive fines. Drying parameters must therefore be controlled carefully to maintain consistent moisture removal without damaging the physical or chemical properties of the formulation.
2. Drying Mechanisms
Pharmaceutical drying processes rely on the transfer of heat to the wet material and the subsequent removal of evaporated solvent through controlled airflow. Two fundamental mechanisms occur during drying:
- Heat transfer from the drying medium to the wet granules
- Mass transfer of evaporated moisture from the granules into the surrounding air stream
Drying typically occurs in two phases: In the initial phase, surface moisture evaporates rapidly as heat is transferred to the wet granule surface. During the later phase, moisture must migrate from the interior of the granule to the surface before evaporation can occur. This internal diffusion stage often determines the overall drying time.
Drying equipment must therefore provide adequate airflow, temperature control, and particle movement to support efficient heat and mass transfer.
3. Fluid Bed Dryers
Fluid bed dryers are the most commonly used drying systems for pharmaceutical wet granulation processes.
In a fluid bed dryer, granules are suspended in an upward flow of heated air. The air velocity is adjusted so that the particles remain fluidized, creating a dynamic particle bed in which granules behave similarly to a boiling liquid. This fluidization provides several advantages:
- Uniform exposure of particles to drying air
- High heat transfer efficiency
- Rapid solvent evaporation
- Uniform moisture distribution within the batch
The drying air is typically filtered and heated before entering the product chamber. As the air passes through the fluidized bed, it removes evaporated solvent and carries it through the exhaust system where particles are separated by filters.
The illustration below shows a typical pharmaceutical fluid bed drying system used to dry wet granules following the granulation step. The system includes the fluid bed dryer vessel, an air handling unit that supplies heated process air, and a filter housing that captures entrained particles before exhaust. During operation, granules are suspended in an upward flow of heated air within the drying chamber, allowing efficient heat transfer and rapid moisture removal while maintaining uniform drying conditions throughout the particle bed.

Fluid bed drying provides efficient and reproducible drying and is widely integrated with fluid bed granulation systems.
4. Tray Dryers
Tray dryers represent a simpler batch drying system used for smaller production volumes or specialized formulations. In tray dryers, wet granules are spread across shallow trays and placed in a heated chamber. Heated air is circulated across the trays to remove moisture from the material surface. Compared with fluid bed drying, tray drying offers:
- Simpler equipment design
- Lower mechanical stress on granules
- Suitability for fragile materials
However, tray drying typically requires longer drying times and may produce less uniform moisture distribution unless trays are rotated or repositioned during drying.
The illustration below shows a tray drying system commonly used in pharmaceutical manufacturing for smaller batches or temperature-sensitive materials. Wet granules are spread across trays and dried by circulating heated air within the drying chamber.

5. Vacuum Dryers
Vacuum dryers remove moisture under reduced pressure conditions. Lower pressure reduces the boiling point of the solvent, allowing drying to occur at lower temperatures. Vacuum drying is particularly useful for:
- Heat-sensitive active pharmaceutical ingredients
- Solvent-based granulation systems
- Materials prone to oxidation
Vacuum drying systems typically consist of a sealed vessel connected to a vacuum pump and heating system. As pressure decreases, solvent evaporation occurs more readily and vapors are removed through the vacuum system.
The illustration below shows a vacuum drying system used for heat-sensitive pharmaceutical materials. Drying occurs under reduced pressure, which lowers the boiling point of the solvent and allows moisture removal at lower temperatures.

6. Critical Design Features of Drying Equipment
Drying equipment must be designed to provide controlled heat transfer, uniform airflow distribution, and reliable moisture removal. Important design features include:
- Uniform airflow distribution across the drying chamber
- Precise temperature control
- Efficient exhaust filtration to capture product particles
- Instrumentation for monitoring airflow, pressure, and temperature
- Access for cleaning and inspection
Fluid bed dryers additionally require properly designed air distribution plates that ensure uniform fluidization of the granule bed. Equipment design must minimize dead zones, channeling of air, and uneven heat distribution that could produce inconsistent drying.
The illustration below demonstrates airflow distribution within a fluid bed dryer. Heated air enters through the distribution plate at the base of the dryer and flows upward through the granule bed, creating fluidization and enabling uniform heat and mass transfer during the drying process.
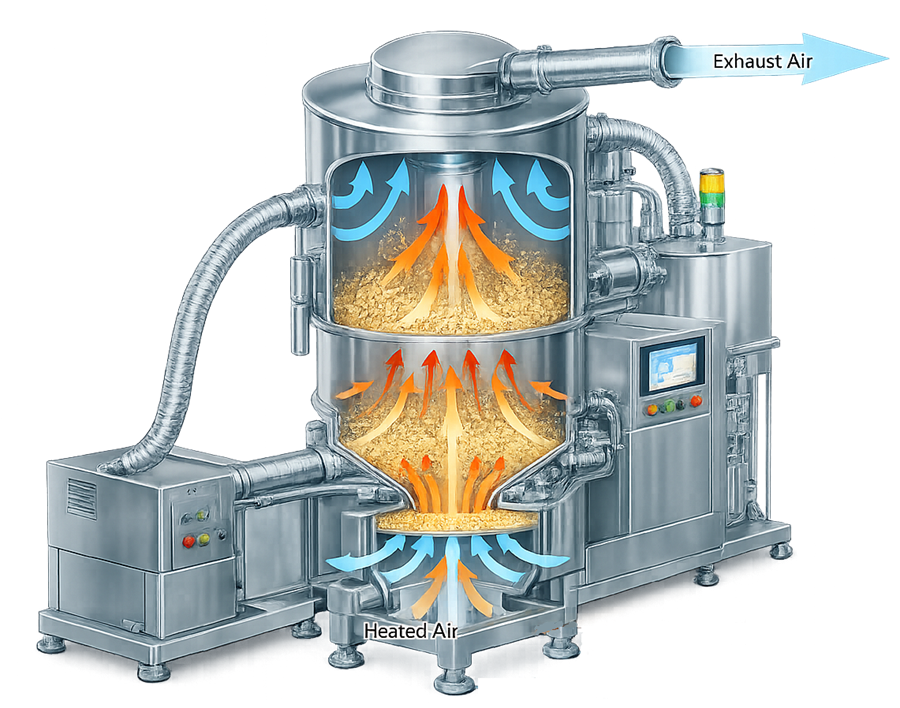
7. Drying Process Monitoring
Drying processes must be monitored to ensure that moisture removal occurs within the validated operating range. Common monitoring parameters include:
- Inlet air temperature
- Outlet air temperature
- Airflow rate
- Exhaust humidity
- Pressure differentials across filters
- Drying time
In many systems, the difference between inlet and outlet air temperature can indicate the progress of drying. As moisture is removed, the outlet temperature gradually approaches the inlet temperature. End-point determination may also rely on direct measurement of residual moisture using sampling and analytical methods.
The illustration below shows a typical drying curve representing moisture reduction during the drying process. The chart presents three curves: granule moisture content, product temperature, and inlet drying air temperature.
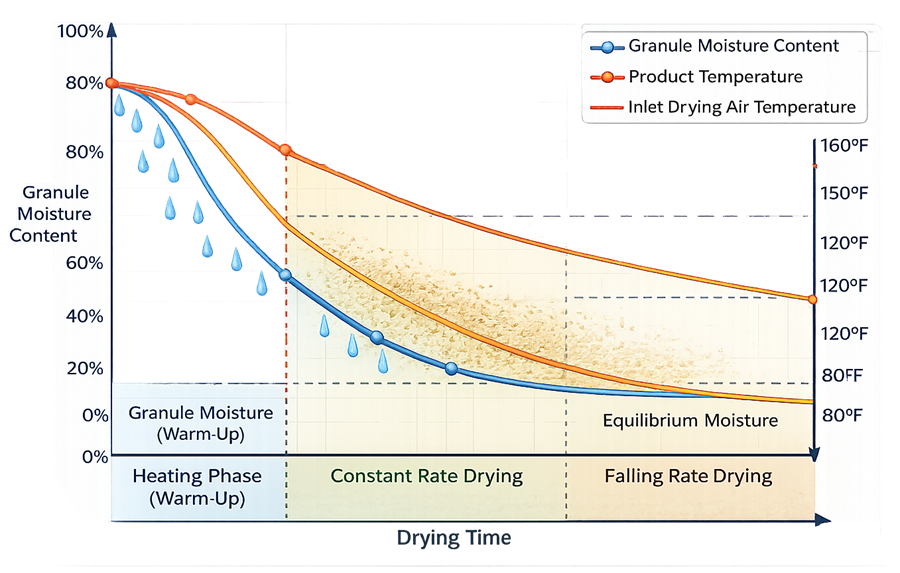
The granule moisture curve shows a continuous decrease as drying progresses, the product temperature curve initially rises, stabilizes during evaporation, and increases again as moisture becomes limited, while the inlet air temperature remains relatively constant because it is controlled by the heating system. The drying process is divided into three phases along the time axis: the heating phase in which granules warm up with limited moisture removal, the constant rate drying phase where surface moisture evaporates rapidly, and the falling rate drying phase in which moisture diffusion from the interior of the granule controls the drying rate.
8. Qualification of Drying Equipment
Drying equipment must undergo formal qualification to demonstrate that the system is installed correctly, operates within defined limits, and supports consistent drying performance. Qualification typically includes:
- Design Qualification
- Installation Qualification
- Operational Qualification
- Performance Qualification
8.1 Design Qualification
Design Qualification verifies that the selected drying equipment meets process and operational requirements defined in the User Requirements Specification. The scope typically includes:
- Equipment capacity relative to batch size
- Airflow distribution design
- Heating system capability
- Exhaust filtration systems
- Instrumentation and control system design
- Materials of construction and surface finish
- Containment and safety features
8.2 Installation Qualification
Installation Qualification confirms that the drying system has been installed in accordance with approved engineering specifications and manufacturer documentation. Installation verification typically includes:
- Equipment identification and model verification
- Mechanical installation verification
- Electrical and control system connections
- Utility connections including compressed air, electrical supply, and heating systems
- Calibration of temperature, pressure, and airflow instrumentation
- Verification of documentation and drawings
8.3 Operational Qualification
Operational Qualification verifies that the drying system operates correctly across its defined operating ranges. Operational testing typically evaluates:
- Airflow control and distribution
- Temperature control accuracy
- Heater performance
- Filter integrity and pressure differential monitoring
- Control system alarms and interlocks
- Safety systems and emergency shutdown functions
Operational tests demonstrate that the equipment can maintain stable operating conditions during drying operations.
8.4 Performance Qualification
Performance Qualification demonstrates that the drying process consistently produces granules meeting predefined moisture specifications under routine manufacturing conditions. PQ confirms that the drying equipment, operating parameters, and process controls operate together to produce stable and reproducible drying performance for the intended formulation and batch size.
PQ execution is typically performed using production-scale equipment and representative batch sizes. Multiple consecutive batches are processed using the validated operating parameters established during process development and Operational Qualification. These batches should reflect routine manufacturing conditions including the intended formulation, batch size, loading configuration, and airflow conditions.
During PQ, drying performance is evaluated by measuring residual moisture content and confirming that moisture distribution remains uniform throughout the batch. The PQ study should also confirm that dried granules perform adequately during downstream operations such as milling, blending, and compression. PQ studies typically evaluate:
- Granule residual moisture content
- Uniformity of moisture across defined sampling locations
- Batch-to-batch reproducibility of drying performance
- Impact on downstream processing performance such as milling and compression behavior
8.4.1 Analytical Methods
Residual moisture measurements must be performed using validated analytical methods capable of accurately quantifying moisture within the expected operating range. Analytical methods used during PQ must demonstrate adequate precision, accuracy, and repeatability. Common analytical methods include:
- Loss on Drying (LOD)
- Karl Fischer moisture analysis
Loss on Drying is commonly used for routine granulation processes and measures the weight loss of a sample during controlled heating. Karl Fischer titration provides higher sensitivity and is often used for low moisture formulations or moisture-sensitive materials. The analytical method selected for PQ should be consistent with the method used during process development and routine quality control testing.
8.4.2 Sampling Strategy
A defined sampling plan is required to evaluate moisture distribution throughout the batch. Sampling locations should represent potential worst-case regions within the drying vessel where heat transfer or airflow distribution may differ. For fluid bed dryers, representative sampling locations may include:
- Top region of the granule bed
- Middle region of the granule bed
- Lower region near the air distribution plate
- Peripheral regions near vessel walls
- Discharge location
The illustration below shows typical sampling locations used during drying process Performance Qualification. Samples are collected from multiple regions of the granule bed to verify uniform moisture removal throughout the batch.
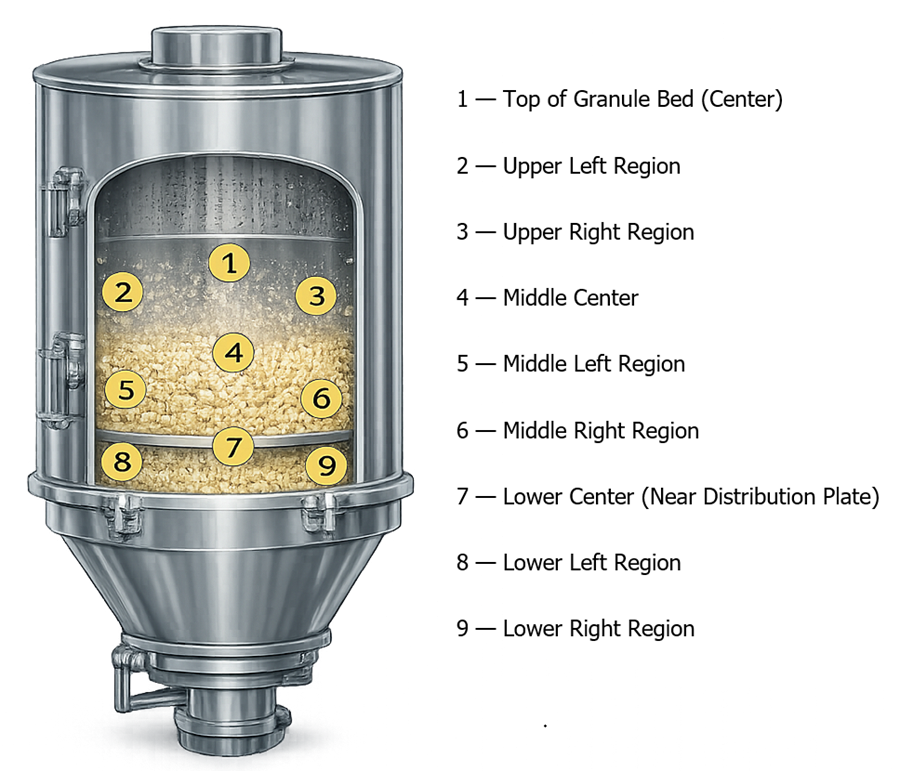
Sampling should be performed at the completion of the drying cycle once the system indicates that the drying end point has been reached. Multiple samples from each location may be collected to ensure that localized moisture variation is detected.
Sampling procedures must ensure that representative samples are obtained without introducing environmental moisture contamination or altering the moisture content of the sample.
8.4.3 Statistical Evaluation
Statistical analysis of PQ data is performed to evaluate drying consistency within each batch and across batches. Moisture measurements collected from multiple sampling locations are analyzed to determine whether the drying process produces uniform and reproducible results. Statistical evaluation typically includes calculation of:
- Mean moisture content
- Standard deviation
- Relative standard deviation
These calculations allow evaluation of variability within a batch and across multiple PQ batches. Comparison of moisture values between sampling locations can confirm whether drying conditions are uniform throughout the granule bed.
Trend evaluation across PQ batches may also be performed to verify that the drying process operates within a stable and controlled operating range. Excessive variability or systematic differences between sampling locations may indicate airflow distribution issues or inadequate drying conditions.
8.4.4 Acceptance Criteria
Acceptance criteria must be defined before PQ execution. Residual moisture content for each sample must fall within the predefined specification limits established during process development. Acceptance criteria should address:
- Moisture content limits for individual samples
- Allowable variability between sampling locations
- Batch-to-batch consistency of drying performance
Moisture values should demonstrate uniform distribution across sampling locations with no evidence of localized under-drying or over-drying. Additionally, the dried granules must demonstrate acceptable performance during downstream operations such as milling, blending, and tablet compression.
Successful completion of Performance Qualification confirms that the drying system consistently produces granules with moisture levels suitable for subsequent processing and supports reliable operation of the validated manufacturing process.
9. Integration with Granulation and Downstream Processing
Drying equipment operates as an intermediate step within the solid dosage manufacturing process. Wet granules produced during granulation are transferred to the dryer where moisture is removed under controlled temperature and airflow conditions. After drying, granules are typically transferred to a sizing mill that breaks large agglomerates and produces a uniform particle size distribution.
The dried granules are then transferred to final blending operations where lubricants and other excipients are added prior to compression or encapsulation. Proper integration between granulation, drying, milling, and blending systems ensures continuous material flow while preventing segregation, contamination, or product loss.
Drying equipment therefore plays a critical role in maintaining consistent granule properties and ensuring reliable downstream processing performance in pharmaceutical solid dosage manufacturing.

