Health-Based Exposure Limits
1. Purpose and Definition
Health-Based Exposure Limits (HBELs) define the maximum daily exposure to a substance that is not expected to cause adverse health effects. In cleaning validation, HBELs establish the scientific basis for acceptable residue levels and control of cross-contamination.
HBELs convert toxicological data into limits that directly determine how clean equipment must be to protect patient safety.
2. Key Terminology
Primary terms used:
- Permitted Daily Exposure (PDE)
maximum acceptable daily intake derived from toxicological data - Acceptable Daily Exposure (ADE)
equivalent to PDE and used interchangeably
Both are expressed in mass per day, typically mg/day.
3. Regulatory and Scientific Basis
HBELs are required under modern risk-based GMP expectations and align with:
- ICH Q9
- ICH Q10
- EMA Guideline on setting health based exposure limits
Regulators expect cleaning limits to be based on compound-specific toxicology rather than fixed or arbitrary criteria.
4. Toxicological Basis
HBELs are derived from toxicological endpoints that define safe exposure levels. Common inputs:
- NOAEL — No Observed Adverse Effect Level
- LOAEL — Lowest Observed Adverse Effect Level
- clinical and pharmacological data
- duration and route of exposure
A scientifically justified point of departure is selected based on data quality and relevance.
5. Calculation Principle
HBEL is calculated by applying adjustment factors to the selected toxicological endpoint:
PDE=F1×F2×F3×F4×F5NOAEL×BW
Where:
- NOAEL = toxicological reference point
- BW = body weight
- F₁–F₅ = uncertainty factors
6. Uncertainty Factors
Adjustment factors account for variability and data limitations.
They address:
- interspecies differences
- human variability
- study duration
- severity of effect
- completeness of data
Selection must be justified and documented.
7. Translation to Cleaning Limits
HBEL is not used directly in validation. It is converted into operational limits:
- Maximum Allowable Carryover
- surface residue limits in µg/cm²
- swab and rinse acceptance criteria
This translation links patient safety to measurable cleaning outcomes.
Health-based exposure limits must be translated into measurable cleaning acceptance criteria. The following diagram shows how toxicological data is converted into operational limits used in cleaning validation.
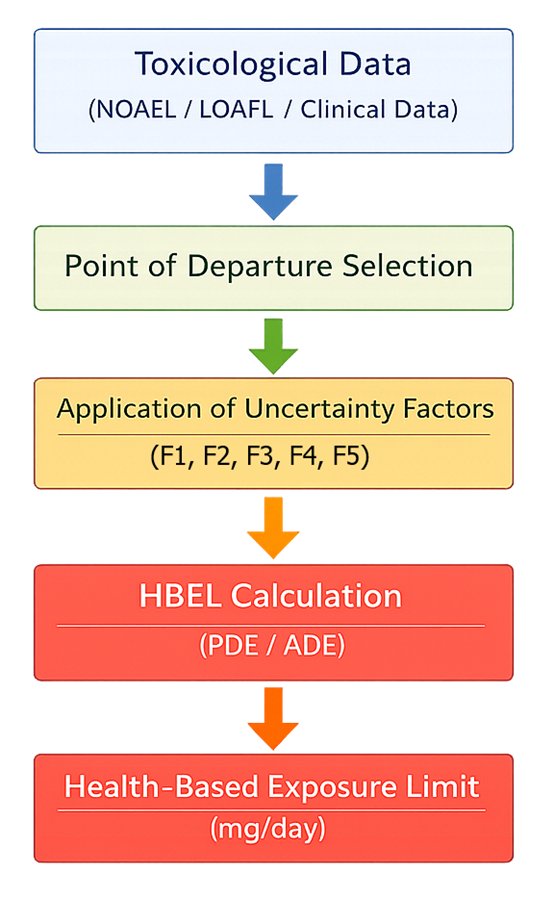
The process begins with toxicological data, including No Observed Adverse Effect Level (NOAEL) and Lowest Observed Adverse Effect Level (LOAEL), as well as clinical and pharmacological data. A point of departure is selected based on the most relevant and reliable data.
This value is then adjusted using uncertainty factors to account for variability and limitations in the data. Each factor modifies the toxicological input to ensure that the resulting exposure limit is protective of patient safety:
- F1 — interspecies differences
animal to human extrapolation - F2 — human variability
differences within the human population - F3 — study duration
adjustment from short-term to long-term exposure - F4 — severity of toxicity
consideration of serious or irreversible effects - F5 — data completeness
compensation for missing or limited data
These factors are applied multiplicatively in the denominator of the calculation, resulting in a conservative and scientifically justified Permitted Daily Exposure (PDE) or Acceptable Daily Exposure (ADE) value.
The final PDE or ADE represents the maximum daily exposure that is not expected to cause adverse effects and serves as the basis for subsequent cleaning validation calculations.
8. Role in Validation Design
HBEL drives key validation decisions:
- worst-case product selection
lower PDE results in stricter limits and higher risk ranking - equipment sharing assessment
low HBEL may require additional controls or segregation - validation stringency
lower HBEL requires tighter limits and more rigorous testing
HBEL defines the level of control required across the validation program.
9. Use in Execution and Evaluation
HBEL-derived limits are used during validation and routine operations:
- evaluation of swab and rinse results against acceptance criteria
- classification of deviations based on patient risk
- confirmation that cleaning processes maintain control
This ensures that results are interpreted in a toxicological context, not only numerically.
10. Documentation Requirements
HBEL determination must be traceable and reviewable.
Required elements:
- source of toxicological data
- selected point of departure
- applied uncertainty factors with justification
- calculation details
- final PDE or ADE value
Typically prepared or approved by a qualified toxicologist.
11. Common Deficiencies
Typical issues:
- use of non–health-based limits without justification
- incorrect or unjustified uncertainty factors
- outdated or non-representative data
- lack of traceability to source data
These deficiencies compromise the scientific validity of cleaning limits.
12. Lifecycle Considerations
HBELs must be maintained over time.
Reassessment is required when:
- new toxicological data becomes available
- products or formulations change
- manufacturing conditions change
HBEL must remain aligned with current knowledge and risk.
13. Key Principle
HBEL defines the acceptable level of exposure.
All cleaning validation limits and decisions are derived from this value.
It is the controlling input that links toxicology to validation and ensures that cleaning processes are scientifically justified and protective of patient safety.

