Incubators and Warm Rooms
1. Overview of Incubators and Warm Rooms
Incubators and warm rooms are controlled thermal environments used to maintain materials, biological cultures, and laboratory samples at defined temperatures above ambient conditions. These systems are widely used in pharmaceutical manufacturing, microbiology laboratories, environmental monitoring programs, and quality control testing operations.

Unlike refrigerated storage equipment designed to remove heat, incubators and warm rooms supply controlled heat in order to maintain stable temperature conditions required for microbial growth, biochemical reactions, and certain stability or incubation studies.
Temperature ranges used in pharmaceutical microbiology are defined by pharmacopeial test methods and the growth requirements of microorganisms being evaluated. Environmental monitoring samples are commonly incubated at 22.5 °C ± 2.5 °C, a range that supports recovery of a broad spectrum of environmental organisms.
Additional incubation temperatures are used for microorganisms that grow optimally near physiological conditions or when specific microbiological methods require elevated temperatures.
Typical incubation ranges used in pharmaceutical laboratories include:
- 20 °C to 25 °C for environmental monitoring and recovery of environmental microorganisms
- 30 °C to 35 °C for microbiological incubation such as sterility testing and growth promotion testing
- 37 °C to 45 °C for specialized biological processes or accelerated microbial growth studies
Incubators typically provide localized controlled environments in benchtop or floor-standing cabinets, while warm rooms provide larger walk-in temperature-controlled spaces for incubation or controlled storage of materials. Both equipment types must maintain stable and uniform temperature conditions to ensure reproducibility of laboratory results and reliable incubation performance.
2. Types of Incubators and Warm Room Systems
Several configurations of thermal incubation equipment are commonly used in regulated pharmaceutical environments.
2.1 Laboratory Incubators
Laboratory incubators are enclosed chambers equipped with controlled heating systems and air circulation to maintain stable internal temperatures. Common types include:
- gravity convection incubators
- mechanical convection incubators with forced air circulation
- CO₂ incubators used for cell culture
- refrigerated incubators capable of operating above and below ambient temperature
The diagram below illustrates the typical internal architecture of a laboratory incubator, including heating elements, air circulation fan, temperature sensors, and the temperature controller used to maintain stable chamber conditions. Mechanical convection incubators distribute heated air through internal airflow circulation, improving temperature uniformity across shelves and reducing temperature gradients within the chamber.
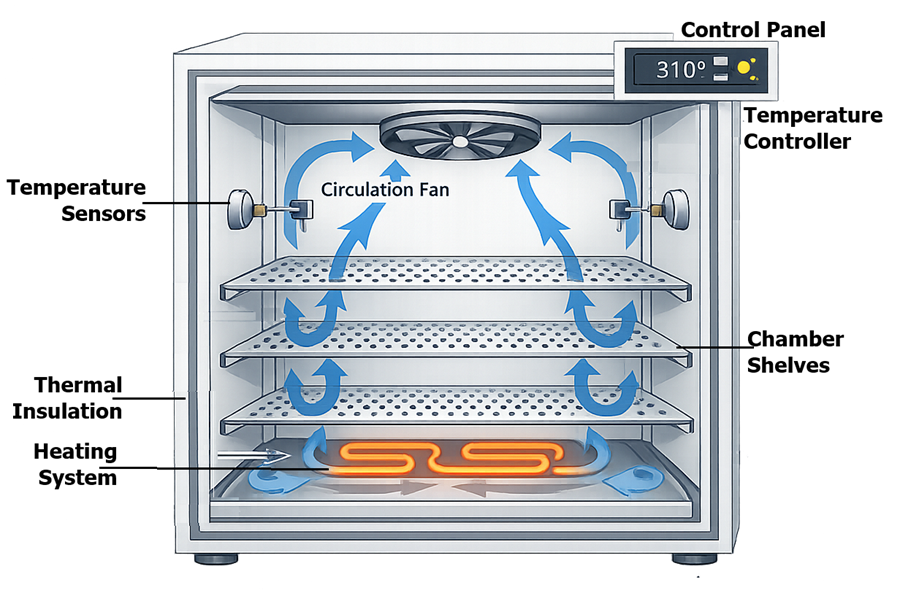
2.2 Warm Rooms
Warm rooms are walk-in temperature-controlled rooms designed to maintain controlled elevated temperatures across a larger storage volume. Warm rooms are typically constructed using insulated panels and dedicated HVAC or heating systems that maintain stable internal temperatures. Temperature control is achieved through:
- electric heating elements
- recirculation fans
- dedicated environmental control systems
These rooms are used when large quantities of materials or equipment must be maintained at elevated temperature conditions. The diagram below illustrates the typical control architecture used to regulate temperature within a walk-in warm room. The system includes heating units, recirculation fans, temperature sensors, and a temperature controller that maintains the defined operating setpoint.
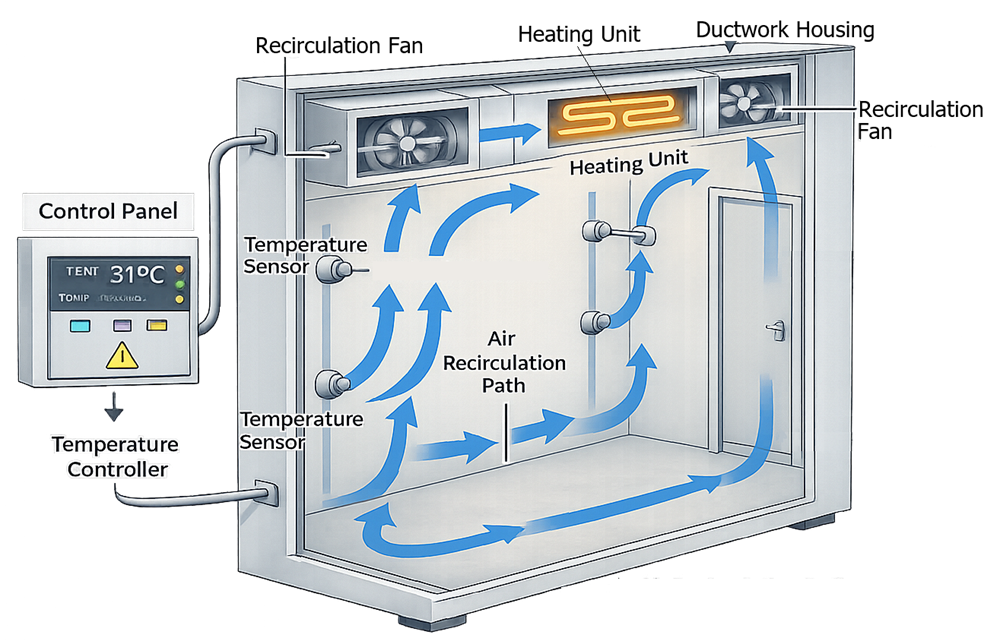
2.3 Specialized Incubation Systems
Certain laboratory processes require specialized incubator configurations including:
- CO₂ incubators for mammalian cell culture
- shaking incubators used for microbial fermentation or suspension cultures
- humidity-controlled incubators
- anaerobic incubation chambers
These systems incorporate additional environmental controls such as gas concentration regulation, agitation mechanisms, or humidity control systems.
3. Temperature Control Architecture
Temperature control in incubators and warm rooms is achieved through integrated heating systems and feedback control loops. Primary system components typically include:
- electric heating elements
- temperature sensors located within the chamber or room
- air circulation fans for temperature distribution
- digital temperature controllers with PID control algorithms
- independent safety thermostats to prevent overheating
Temperature sensors continuously measure internal temperature conditions. The control system adjusts heater output to maintain the programmed setpoint.
Forced air circulation systems are commonly used to distribute heat evenly within the chamber. Without adequate air circulation, localized temperature gradients may develop.
3.1 Airflow and Heat Distribution
Airflow patterns inside incubators strongly influence temperature uniformity within the chamber. Proper air circulation distributes heat throughout the internal space and minimizes localized temperature differences between shelves or chamber regions.
Mechanical convection incubators use internal circulation fans to move heated air across shelves and chamber walls. Continuous air movement promotes uniform temperature distribution, reduces temperature stratification, and improves temperature recovery following door openings.
Warm rooms apply the same principle at a larger scale. Ceiling-mounted or wall-mounted recirculation fans continuously move air throughout the room volume, ensuring that heated air is distributed evenly and that temperature conditions remain stable across the entire controlled space.
3.2 Control System Monitoring
Most regulated laboratory incubators include digital monitoring systems capable of:
- continuous temperature monitoring
- alarm generation for out-of-range conditions
- data logging of temperature history
Alarm systems typically include:
- high temperature alarms
- low temperature alarms
- door ajar alarms indicating that the chamber door has remained open beyond a defined time
- sensor failure detection
- power interruption alarms
Specialized incubators may include additional environmental monitoring alarms such as:
- high and low CO₂ concentration alarms for CO₂ incubators
- high and low humidity alarms for humidity-controlled incubation systems
These alarms ensure that deviations are detected promptly and that incubation parameters remain within defined operating limits, supporting environmental monitoring programs and data integrity requirements in regulated environments.
4. Operational Considerations
Proper operation of incubators and warm rooms is required to maintain stable temperature conditions. Operational practices typically include:
- minimizing door openings to reduce temperature fluctuations
- avoiding overloading shelves that restrict airflow
- maintaining adequate spacing between stored items
- ensuring air vents and circulation pathways remain unobstructed
Frequent door openings introduce ambient air into the chamber and can temporarily disrupt temperature stability. Large loads with high thermal mass may also influence recovery time after temperature disturbances.
4.1 Temperature Recovery Behavior
After a door opening event, incubators must return to the defined temperature setpoint within an acceptable recovery time. Recovery performance depends on several factors:
- heater capacity
- air circulation efficiency
- chamber volume
- thermal mass of stored materials
Stable recovery characteristics are important for processes that require consistent incubation conditions.
The diagram below illustrates the typical temperature disturbance and recovery behavior following a door opening event. When the door is opened, ambient air enters the chamber and causes a temporary temperature deviation. The system then restores the chamber temperature to the defined setpoint through heater operation and internal air circulation.
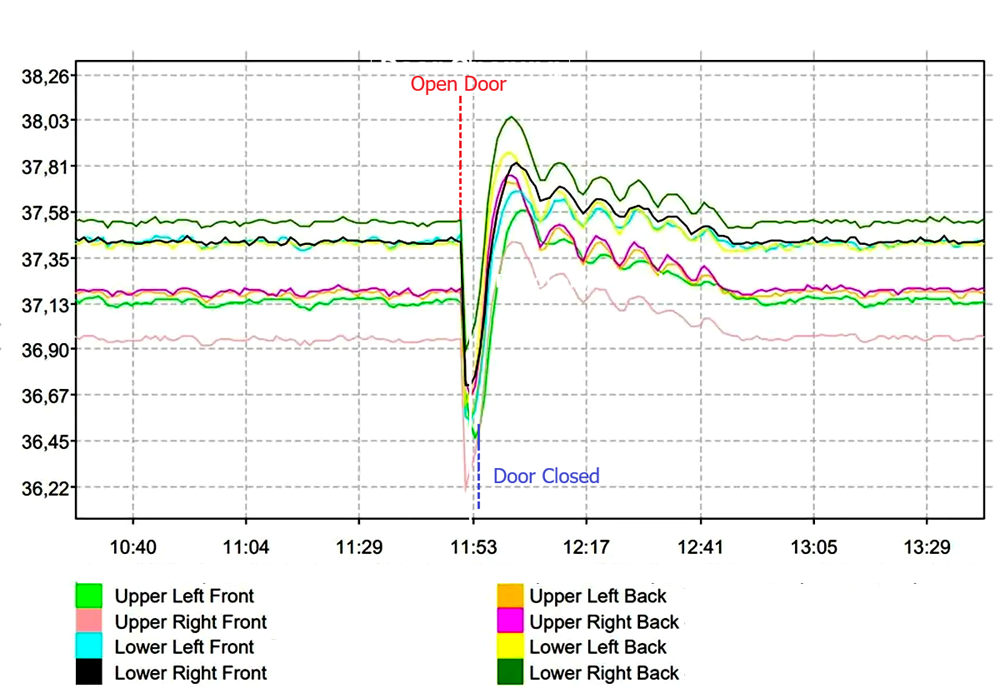
5. Qualification and Validation Considerations
Incubators and warm rooms used in regulated pharmaceutical environments must be qualified to demonstrate that temperature conditions are maintained within defined operating limits during routine operation. Qualification activities confirm that the equipment is capable of maintaining the required incubation conditions and that monitoring and safety systems function as intended. Typical qualification testing includes verification of:
- temperature control accuracy
- temperature stability over time
- alarm functionality
- temperature recovery following door openings or other disturbances
In addition to functional testing, temperature distribution studies are performed to demonstrate that uniform temperature conditions are maintained throughout the chamber or room. These studies evaluate temperature variation across different locations within the controlled space and confirm that all monitored positions remain within the defined operating range.
Temperature stratification can occur in incubators, particularly in systems that rely on gravity convection rather than forced air circulation. Warm rooms may also exhibit temperature gradients due to room size, airflow patterns, and heat distribution within the space. For this reason, qualification studies must verify that temperature differences across the chamber or room remain within acceptable limits under normal operating conditions.
These studies establish the equipment’s operational capability and confirm that the incubation environment remains suitable for microbiological and laboratory applications.
5.1 Sensor Placement and Monitoring
Incubators and warm rooms typically incorporate several types of temperature sensors that serve different operational and monitoring functions. These sensors are used for temperature control, operational monitoring, alarm generation, and documentation of environmental conditions.
The primary control sensor is connected to the temperature controller and provides the feedback signal used by the control system to regulate heater output and maintain the defined setpoint. Accurate positioning of the control sensor is important because it directly influences the temperature regulation performance of the system.
In addition to the control sensor, many systems include independent monitoring sensors that provide secondary measurement of chamber conditions. These sensors may be connected to digital displays, alarm systems, environmental monitoring systems, or building monitoring infrastructure.
In regulated laboratory environments, temperature monitoring may also include independent data acquisition devices used for documentation and trending of incubation conditions. These systems may include:
- independent monitoring probes connected to environmental monitoring systems
- stand-alone temperature data loggers used for continuous monitoring
- chart recorders used for graphical recording of temperature history
- SCADA or building management system interfaces for centralized monitoring
For large chambers or warm rooms, multiple monitoring probes may be installed at representative locations to provide more accurate assessment of the controlled environment. Sensor placement is typically selected to represent the general chamber conditions while avoiding direct exposure to heating elements or airflow outlets that may create localized temperature effects.
5.2 Alarm and Safety Systems
Incubators and warm rooms are equipped with alarm and protective safety systems intended to detect abnormal operating conditions and prevent damage to stored materials or equipment. These systems provide notification when environmental parameters deviate from defined limits and help ensure that incubation conditions remain within the validated operating range.
Alarm functions are typically integrated into the temperature control system and generate audible, visual, or electronic notifications when predefined limits are exceeded or when equipment faults occur. Alarm signals may be displayed locally on the equipment interface or transmitted to centralized monitoring systems used for laboratory environmental monitoring.
In addition to alarm notification, most incubators include independent safety devices designed to prevent overheating or uncontrolled heating conditions. These protective devices operate separately from the primary temperature controller and automatically interrupt heater operation if unsafe temperature conditions are detected.
Typical protective mechanisms include independent over-temperature safety thermostats, secondary temperature limit controllers, and automatic heater shutdown circuits. These systems provide a fail-safe response in the event of controller malfunction, sensor failure, or other abnormal conditions that could cause excessive temperature rise.
Verification of alarm functionality and safety system performance is an important component of equipment qualification. Testing typically confirms that alarm conditions are detected correctly and that protective devices respond appropriately to simulated fault conditions.
6. Applications in Pharmaceutical Operations
Incubators and warm rooms are used in pharmaceutical laboratories and manufacturing support areas where controlled elevated temperature conditions are required for microbiological or biological processes. These systems provide stable thermal environments that support microbial growth, culture incubation, and controlled laboratory studies.
In pharmaceutical quality control laboratories, incubators are commonly used for microbiological testing activities. Typical applications include incubation of environmental monitoring samples, sterility testing, and growth promotion testing of microbiological culture media. These activities require defined temperature conditions to ensure reliable recovery and growth of microorganisms.
Warm rooms may be used when larger quantities of materials must be maintained at controlled elevated temperatures. Examples include storage of prepared microbiological media, incubation of large batches of environmental monitoring plates, or temporary storage of biological materials requiring controlled warm conditions.
Because microbial growth rates and biological reactions are strongly influenced by temperature, stable and uniform thermal conditions are necessary to ensure reproducible laboratory results and consistent performance of microbiological test methods.

