Maximum Allowable Carryover
1. Purpose and Definition
Maximum Allowable Carryover (MACO) defines the maximum quantity of a product residue that may be carried over from one manufacturing batch to the next without posing a risk to patient safety or product quality.
MACO establishes the numerical acceptance criteria used in cleaning validation. It converts toxicological limits into measurable residue limits on equipment surfaces or in rinse solutions.
2. Regulatory and Scientific Basis
MACO calculations are expected under 21 CFR 211.67 and must be supported by scientifically justified limits derived from toxicological data.
Modern regulatory expectations align with:
- ICH Q9
- ICH Q10
Health-based exposure limits such as Permitted Daily Exposure or Acceptable Daily Exposure are the preferred basis for MACO determination.
3. Conceptual Model
MACO is based on the principle that any residue carried over into the next product must not result in patient exposure exceeding the allowable daily intake.
The calculation links:
- toxicity of the previous product
- dose and batch size of the next product
- distribution of residue across equipment
This ensures that cleaning limits are directly tied to patient safety.
4. MACO Calculation Approach
4.1 Health-Based MACO Equation
The standard approach uses a health-based exposure limit:
MACO=TDDnextPDE×MBSnext
Where:
- PDE = permitted daily exposure of the previous product
- MBSₙₑₓₜ = minimum batch size of the next product
- TDDₙₑₓₜ = total daily dose of the next product
This equation ensures that residue carried into the next batch will not exceed the acceptable daily exposure for a patient.
Maximum allowable carryover must be translated from a toxicological limit into measurable cleaning acceptance criteria. The following diagram illustrates how MACO is converted into surface, swab, and rinse limits used during cleaning validation.
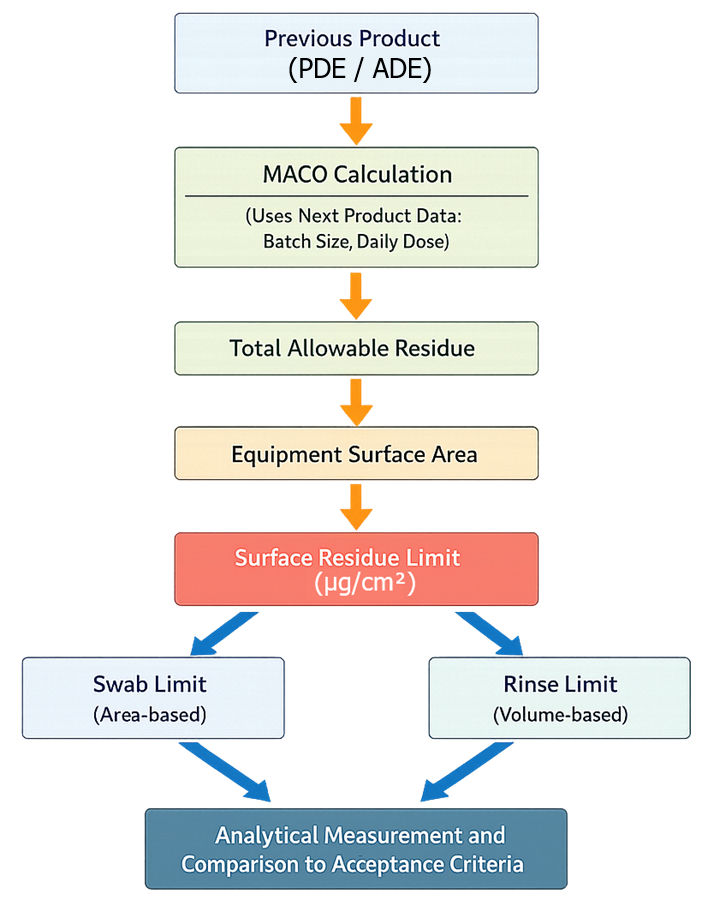
The calculation begins with the toxicological limit of the previous product, expressed as permitted or acceptable daily exposure. This value is used together with the batch size and dosing parameters of the next product to determine the total allowable residue that can be carried over.
This total residue is then distributed across the total shared equipment surface area to establish a surface limit. The surface limit is further converted into swab and rinse limits based on sampling area and rinse volume. These limits define the acceptance criteria against which analytical results are evaluated during cleaning validation.
4.2 Conversion to Surface Limit
MACO must be converted into a measurable limit for cleaning validation.
Surface Limit=AtotalMACO
Where:
- Aₜₒₜₐₗ = total shared equipment surface area
This provides a residue limit expressed as mass per unit surface area, typically µg/cm².
4.3 Conversion to Swab or Rinse Limits
For practical testing:
- swab limits are calculated based on sampled surface area
- rinse limits are calculated based on rinse volume
These conversions ensure that analytical results can be directly compared to acceptance criteria.
5. Alternative Approaches
Where toxicological data is limited, alternative methods may be used:
- Dose-based approach
using a fraction of the minimum therapeutic dose - 10 ppm criterion
limiting carryover to 10 parts per million in the next product
These approaches are considered less scientifically robust and should be justified when used.
6. Worst-Case Considerations
MACO calculations must reflect worst-case conditions:
- lowest PDE of previous product
- smallest batch size of next product
- highest daily dose of next product
Failure to use worst-case parameters results in non-conservative limits.
7. Application in Cleaning Validation
MACO defines the acceptance criteria for:
- swab samples from equipment surfaces
- rinse samples from process systems
Cleaning validation is successful only when all measured residues are below calculated limits.
8. Documentation Requirements
MACO calculations must be fully documented and traceable.
Required elements include:
- source of Permitted Daily Exposure (PDE) or Acceptable Daily Exposure (ADE) values
- calculation inputs and assumptions
- batch size and dose justification
- surface area determination
- conversion to analytical limits
Documentation must allow independent verification of the calculation.
9. Common Deficiencies
Typical failures include:
- use of non–health-based limits without justification
- incorrect batch size or dose assumptions
- inconsistent surface area calculations
- failure to apply worst-case conditions
- lack of traceability to toxicological data
These deficiencies undermine the scientific validity of cleaning limits.
10. Lifecycle Management
MACO must be maintained throughout the product lifecycle.
Reassessment is required when:
- new products are introduced
- toxicological limits are updated
- equipment configuration changes
- cleaning procedures are modified
MACO is not static and must remain aligned with current manufacturing and safety data.

