Method Specificity and Selectivity
1. Purpose and Definition
Specificity and selectivity define the ability of an analytical method to measure the target residue accurately in the presence of other substances. In cleaning validation, this ensures that measured results represent the actual residue of interest and are not affected by interferences.
- Specificity: ability to measure a single analyte unequivocally
- Selectivity: ability to distinguish the analyte from other components in the matrix
Both are critical for reliable residue determination.
2. Role in Cleaning Validation
Analytical methods must demonstrate that they can:
- detect product residues in the presence of cleaning agents
- distinguish residues from excipients and formulation components
- avoid interference from swab materials and rinse solvents
- provide accurate results under actual sampling conditions
Without specificity and selectivity, analytical results cannot be trusted.
3. Sources of Interference
Cleaning validation samples are complex and may contain multiple interfering substances.
Typical sources include:
- cleaning agents
detergents, solvents, or disinfectants - excipients
binders, oils, surfactants, polymers - degradation products
formed during processing or cleaning - sampling materials
extractables from swabs or containers - matrix components
rinse solvents or extraction solutions
These components may produce signals that overlap with the target analyte.
4. Evaluation of Specificity and Selectivity
Specificity and selectivity must be demonstrated experimentally.
Typical evaluations include:
- analysis of blank samples
confirm absence of interfering peaks or signals - analysis of individual components
assess contribution of cleaning agents and excipients - spiked samples
confirm accurate detection of analyte in presence of matrix - comparison of chromatographic profiles
verify separation of analyte from other components
The method must clearly distinguish the analyte signal from all potential interferences.
Specificity and selectivity are demonstrated by the ability of the analytical method to distinguish the analyte signal from interfering components.
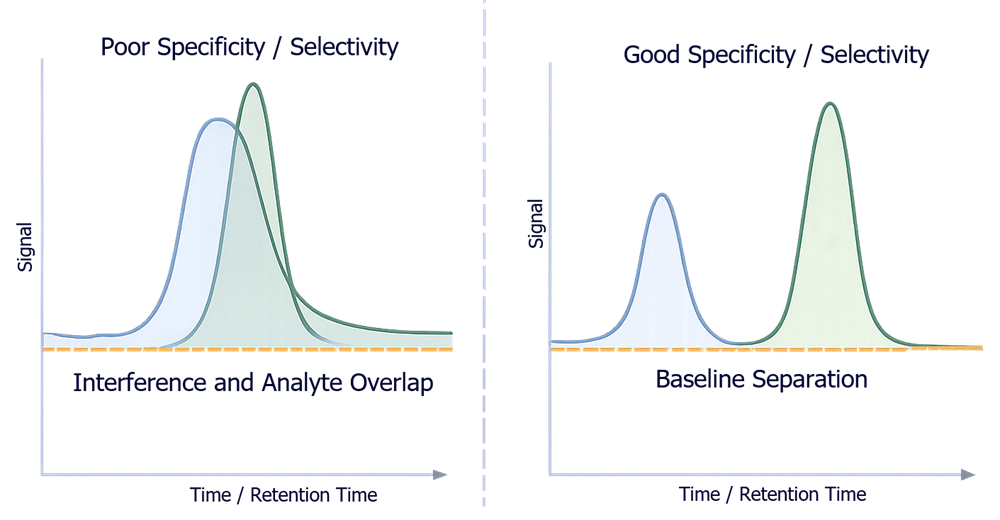
Inadequate specificity results in overlapping signals where the analyte cannot be reliably identified or quantified. Adequate specificity is achieved when the analyte peak is clearly separated from all other components, allowing accurate measurement without interference.
5. Chromatographic Methods
For techniques such as HPLC:
- analyte peak must be well resolved
- no co-eluting peaks at analyte retention time
- peak purity must be confirmed where applicable
Chromatographic separation is the primary mechanism for achieving specificity.
6. Non-Specific Methods
Methods such as Total Organic Carbon (TOC) or conductivity are inherently non-specific.
Characteristics:
- measure total signal rather than individual compounds
- cannot distinguish between different residues
- sensitive to all organic or ionic components
Use of non-specific methods requires:
- justification of applicability
- control of background contributions
- demonstration that signal represents worst-case residue
7. Matrix Effects
Matrix effects occur when sample components influence analytical response.
Examples:
- suppression or enhancement of signal
- altered extraction efficiency
- interference with detection
Matrix effects must be evaluated using representative swab extracts and rinse samples.
8. Acceptance Criteria
Acceptance criteria must ensure that:
- no significant interference occurs at analyte signal
- analyte can be accurately measured in presence of matrix
- method performance is consistent across conditions
Criteria may include:
- resolution between peaks
- absence of signal in blanks
- acceptable recovery in spiked samples
9. Documentation Requirements
Specificity and selectivity must be documented.
Required elements include:
- description of potential interferences
- experimental data demonstrating absence of interference
- chromatograms or analytical outputs
- justification for method selection
Documentation must support independent review.
10. Common Deficiencies
Typical issues include:
- failure to evaluate cleaning agent interference
- inadequate assessment of swab extractables
- reliance on non-specific methods without justification
- unresolved peaks in chromatographic methods
- lack of matrix-based evaluation
These deficiencies compromise analytical reliability.
11. Lifecycle Considerations
Specificity and selectivity must be maintained over time.
Reassessment is required when:
- new products or cleaning agents are introduced
- analytical methods are modified
- sampling materials change
Method suitability must remain aligned with current conditions.
12. Key Principle
Specificity and selectivity ensure that what is measured is truly the target residue.
Without them, analytical results may reflect interference rather than actual contamination.

