Package Types
1. Purpose
This section defines the primary types of packaging used in pharmaceutical applications and their functional roles in protecting product quality during storage and distribution.
2. Primary Packaging
Primary packaging is in direct contact with the product and is critical to maintaining product quality and stability. Selection is driven by product characteristics and administration route.
Common types include:
- vials and ampoules for sterile liquids
- prefilled syringes and cartridges
- bottles for liquids and solids
- blister packs for solid oral dosage forms
- flexible bags for solutions and biologics
Primary packaging must ensure compatibility, prevent contamination, and maintain container closure integrity.
The diagram below shows the hierarchical structure of pharmaceutical packaging and the interaction of temperature-controlled systems across all packaging levels.
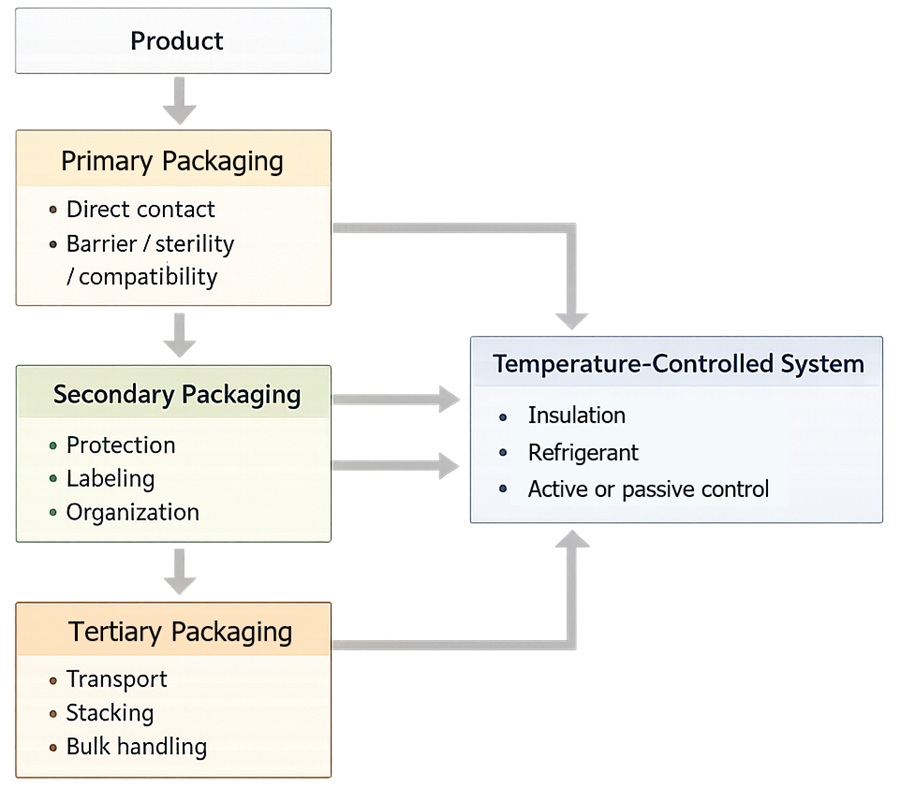
3. Secondary Packaging
Secondary packaging provides additional protection and supports labeling, identification, and handling.
Typical forms include:
- cartons and boxes
- trays and inserts
- protective sleeves or wraps
Secondary packaging protects primary containers from mechanical damage and ensures proper organization during handling and distribution.
4. Tertiary Packaging
Tertiary packaging is used for bulk handling, storage, and transport.
Common configurations include:
- corrugated shipping cases
- pallets and palletized loads
- stretch wrap and strapping systems
Tertiary packaging must withstand stacking, compression, and transport-related stresses.
5. Temperature-Controlled Packaging
Temperature-controlled systems are used for products requiring defined environmental conditions.
Types include:
- passive systems using insulation and refrigerants
- active systems with powered temperature control
Design must ensure maintenance of required temperature ranges throughout distribution.
6. Special Packaging Systems
Certain products require specialized packaging to address specific risks.
Examples include:
- sterile barrier systems for aseptic products
- light-protective packaging for photosensitive products
- tamper-evident packaging
- high-barrier systems for oxygen- or moisture-sensitive products
7. Selection Considerations
Selection of package type must be based on product requirements, distribution conditions, and regulatory expectations. The chosen system must provide adequate protection against environmental exposure, mechanical stress, and contamination while supporting labeling, traceability, and handling requirements.

