Packaging Design Considerations
1. Purpose
This section defines the key factors that must be evaluated when designing packaging systems to ensure protection of pharmaceutical products throughout storage and distribution. Design outputs establish the basis for packaging qualification and shipping validation.
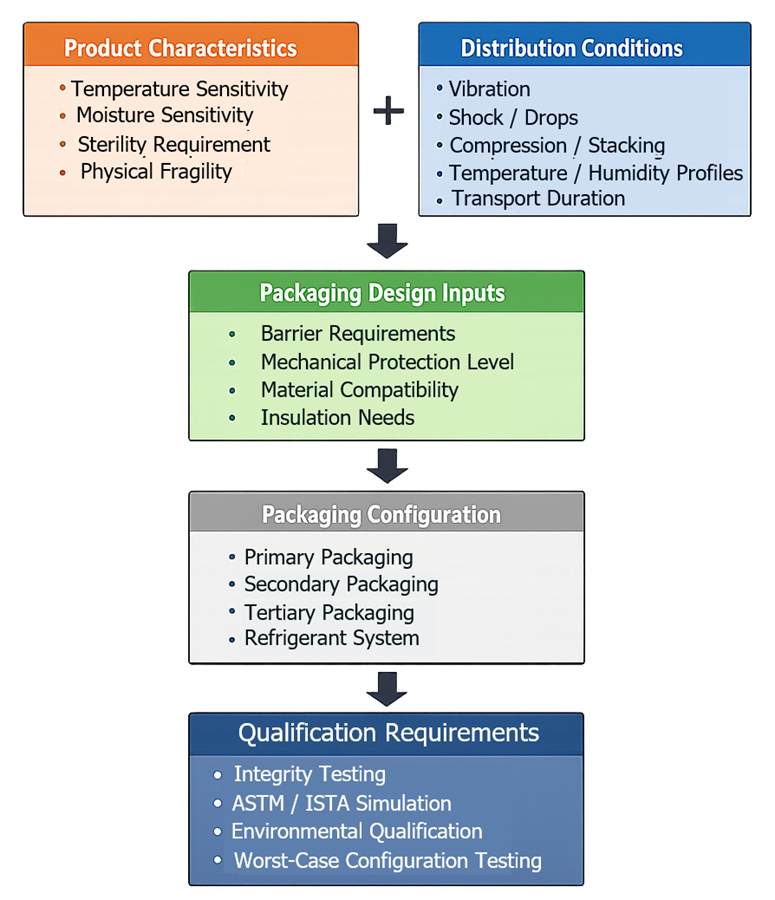
The diagram below illustrates how product and distribution inputs drive packaging design and qualification requirements.
2. Regulatory Expectations
Packaging design must ensure product quality is maintained through the distribution lifecycle in accordance with:
- 21 CFR Part 211, including §211.94 for container-closure systems
- EU GMP Annex 1 for sterile product protection
- ICH Q1A(R2) for environmental sensitivity and stability considerations
- ICH Q9 for risk-based design
Packaging must be suitable for its intended use and must not alter product safety, identity, strength, quality, or purity.
3. Product Characteristics
Packaging design must be driven by product-specific attributes, including:
- sensitivity to temperature, humidity, light, and oxygen
- physical form such as liquid, solid, or lyophilized
- sterility requirements and contamination risk
- fragility and susceptibility to mechanical damage
These characteristics define required barrier properties, protection level, and configuration.
4. Material Selection
Materials must be compatible with the product and suitable for intended conditions of use:
- evaluation of extractables and leachables
- moisture vapor transmission rate and oxygen permeability
- light transmission properties
- chemical inertness and stability
Materials must not interact with the product or compromise quality over shelf life.
5. Packaging Configuration
Packaging design must define the complete system:
- primary packaging such as vials, syringes, bottles, or blisters
- secondary packaging for additional protection and labeling
- tertiary packaging for shipping and handling
Configuration must ensure mechanical protection against shock, vibration, and compression, and support consistent assembly and handling.
6. Distribution Considerations
Packaging design must incorporate expected distribution conditions:
- transport duration and route variability
- handling practices and stacking conditions
- vibration and shock exposure
- environmental conditions including temperature and humidity
These inputs define worst-case conditions for qualification and testing.
7. Container Closure Integrity
For sterile and sensitive products, packaging must maintain integrity throughout lifecycle:
- prevention of microbial ingress
- resistance to leakage under pressure and handling stress
- maintenance of seal integrity during storage and transport
Integrity requirements must be defined and verified during qualification.
8. Labeling and Compliance
Packaging must support regulatory and operational requirements:
- labeling legibility and durability
- traceability and identification
- tamper evidence where applicable
Labeling must remain intact and readable throughout distribution.
9. Design Output
The outcome of packaging design considerations is a defined and justified packaging system including materials, configuration, and performance requirements. These outputs establish acceptance criteria and test conditions for packaging qualification and distribution validation.

