Control Strategy Definition and Lifecycle Management
1. Purpose
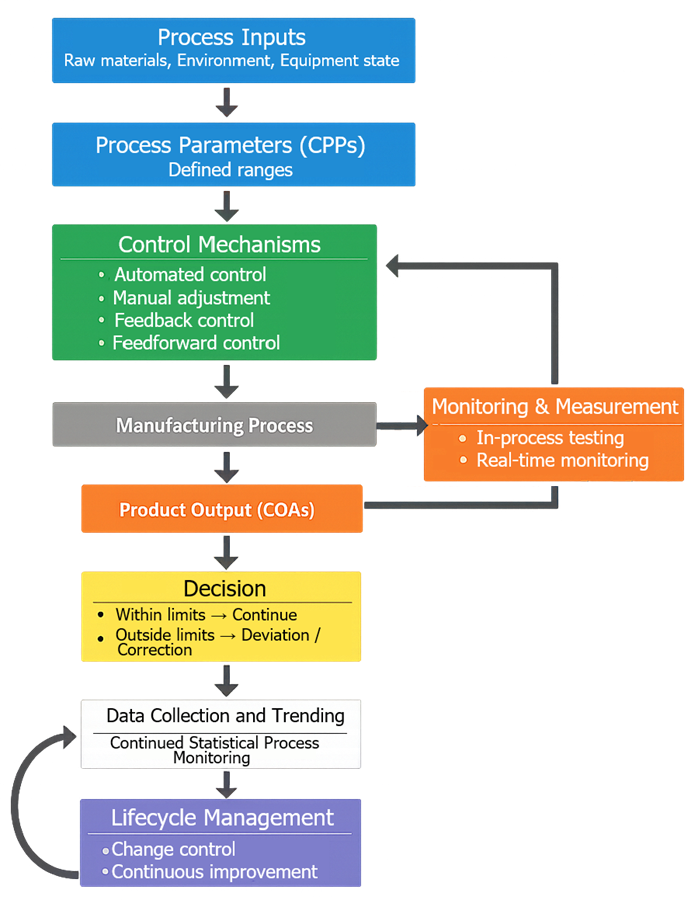
Control strategy defines how process knowledge is translated into operational control during manufacturing to ensure consistent product quality. It establishes how CPPs are controlled to maintain CQAs within defined limits under routine production conditions.
This article defines how control strategy is implemented in manufacturing, how control effectiveness is verified during PPQ, and how performance is maintained through continued process verification and change management.
2. Inputs to Control Strategy
Control strategy is derived from process understanding and risk assessment. Key inputs:
- identified CQAs
- identified CPPs linked to CQAs
- acceptable operating ranges
- process development and characterization data
- identified sources of variability
These inputs define what must be controlled and within what limits.
3. Control Implementation Model
Control strategy defines how process variables are actively managed during manufacturing. It establishes:
- how parameters are set and maintained
- where controls are applied in the process
- how deviations are detected
- how corrective actions are triggered
Control mechanisms include:
- automated control maintaining setpoints
- manual adjustment within defined ranges
- feedback control based on measured outputs
- feedforward control based on input variability
4. Types of Controls
Control strategy consists of layered controls across materials, process, and equipment. Primary control categories:
- Parameter Controls
- defined limits for CPPs
- monitoring and adjustment mechanisms
- In-Process Controls
- sampling during manufacturing
- testing of intermediate material
- Real-Time Monitoring
- continuous measurement of parameters
- automated data capture
- Material Controls
- raw material specifications
- supplier qualification and incoming verification
- Equipment Controls
- defined system settings
- calibration and maintenance
- alarms and interlocks
Each control must define:
- what is controlled
- how it is controlled
- acceptable limits
- monitoring method or frequency
- required actions when limits are exceeded
5. Integration into Manufacturing Operations
Control strategy is executed through manufacturing systems. Implementation elements:
- batch records defining parameter ranges and required actions
- SOPs defining control procedures and responses
- automation systems enforcing limits and alarms
- operator instructions and training
Controls must be executable in real time and consistently applied.
6. Control Verification During PPQ
PPQ evaluates control strategy under commercial conditions. Objectives:
- confirm CPPs remain within defined ranges
- verify monitoring and control systems function correctly
- demonstrate CQAs meet acceptance criteria
- evaluate performance under normal variability
Control failures must be investigated and corrected.
7. Control Performance During Continued Process Verification
Control performance is evaluated using routine manufacturing data. Activities:
- monitoring adherence to parameter limits
- trending CPPs and CQAs
- evaluating control effectiveness
- detecting shifts or trends
Defined limits include:
- normal operating ranges
- alert limits
- action limits
8. Change Management of Control Strategy
Control strategy is updated based on process knowledge and performance. Continued Process Verification relies on structured analysis of manufacturing data to confirm that the process remains in a state of control. This includes routine evaluation of CPPs and CQAs using statistical methods to detect variability, trends, and potential loss of control before product quality is impacted.
Triggers:
- process improvements
- equipment or material changes
- deviations or failures
- identified trends
All changes must be:
- evaluated for impact on CQAs
- supported by data and risk assessment
- implemented through change control
- verified where required
9. Documentation and Traceability
Control strategy must be documented in operational form. Documentation includes:
- defined CPP ranges and limits
- monitoring and testing requirements
- control methods and response actions
- integration into batch records and SOPs
Traceability must demonstrate:
- linkage from process understanding to controls
- verification during PPQ
- ongoing confirmation during routine manufacturing

