Process Validation Documentation Structure
1. Purpose
Process validation documentation and deliverables define the structured set of records used to plan, execute, evaluate, and maintain validation activities. They ensure that validation is traceable, reproducible, and inspection-ready, and that all conclusions are supported by documented evidence.
2. Role Within the Validation Lifecycle
Documentation spans all stages of process validation and provides continuity between development, qualification, and routine manufacturing. Key roles:
- define intended process and acceptance criteria
- document execution of qualification activities
- capture objective evidence of performance
- support decision-making and release
- maintain traceability across lifecycle stages
Documentation is not a byproduct. It is the mechanism by which validation is demonstrated.
3. Core Documentation Structure
Process validation documentation is organized into defined document types with specific roles. Primary structure:
- Validation Plan
- defines scope, strategy, responsibilities, and acceptance criteria
- Risk Assessment
- identifies CPPs, CQAs, and sources of variability
- defines control strategy and sampling approach
- Protocols
- define execution steps, data collection, and acceptance criteria
- include PPQ protocols and supporting test plans
- Raw Data and Records
- manufacturing data
- laboratory results
- monitoring data
- batch records
- Reports
- summarize execution
- evaluate results against acceptance criteria
- provide conclusions
- Continued Process Verification Records
- trending data
- monitoring reports
- periodic reviews
The following diagram illustrates the structure of process validation documentation and how information flows from planning through execution and reporting into continued process verification. It highlights the linkage between documents and the role of traceability in supporting validation conclusions.
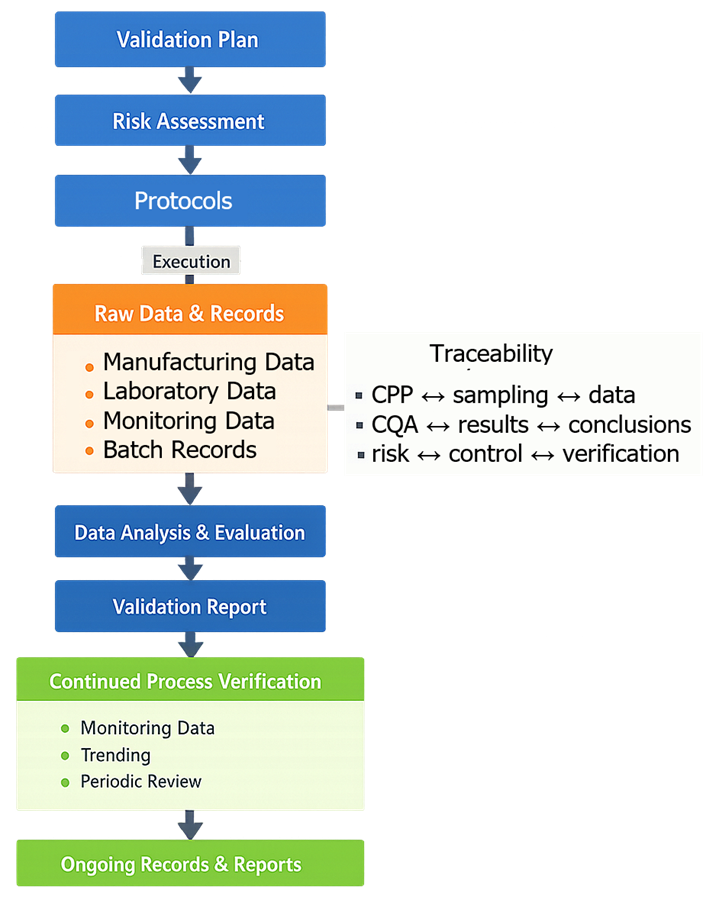
4. Validation Plan
The validation plan defines how process validation will be executed. Content includes:
- scope and process description
- validation approach and lifecycle stages
- identification of CPPs and CQAs
- sampling strategy and statistical methods
- acceptance criteria for PPQ
- roles and responsibilities
The plan establishes the framework for all subsequent documentation.
5. Protocols
Protocols define how validation activities are executed. Key elements:
- defined test steps and sequence
- data collection requirements
- sampling locations and frequency
- acceptance criteria linked to CPPs and CQAs
- handling of deviations
Protocols must be executable and aligned with actual manufacturing operations.
6. Data and Evidence Collection
Validation relies on complete and accurate data collected during execution. Data types:
- process parameter data
- in-process testing results
- finished product testing
- environmental and utility data
- operator records and confirmations
All data must be:
- attributable to specific batches
- complete and consistent
- traceable to original records
7. Reporting and Evaluation
Validation reports document results and conclusions. Report content:
- summary of execution
- evaluation of data against acceptance criteria
- assessment of process variability and consistency
- identification of deviations and investigations
- justification of conclusions
Reports must clearly demonstrate whether the process is in a state of control.
8. Traceability and Linkage
Documentation must provide clear linkage between all elements of validation. Traceability must demonstrate:
- linkage from risk assessment to CPPs and CQAs
- linkage from CPPs to sampling and monitoring
- linkage from data to acceptance criteria
- linkage from results to conclusions
Traceability ensures that validation is scientifically justified and auditable.
9. Continued Process Verification Documentation
Ongoing lifecycle activities require continued documentation. Key records:
- routine monitoring data
- statistical trending outputs
- periodic review reports
- investigation and deviation records
These records demonstrate that the validated state is maintained.
10. Document Control and Governance
All validation documentation must be controlled within the quality system. Requirements:
- version control and approval
- defined ownership and responsibilities
- controlled distribution and access
- retention in accordance with regulatory requirements
Changes to documentation must be managed through formal change control.
11. Inspection Readiness
Validation documentation must support regulatory inspection and internal audit. Expectations:
- complete and organized documentation set
- clear linkage between documents and data
- availability of raw data and supporting evidence
- consistency between reported results and source data
Documentation must demonstrate that validation was planned, executed, and maintained in a controlled and compliant manner.

