General Principles of Process Validation
1. Purpose
General principles of process validation define the foundational concepts that govern how manufacturing processes are designed, qualified, and maintained to ensure consistent product quality.
This article aligns with the principles established in the U.S. Food and Drug Administration Process Validation guidance and focuses on how those principles are applied in practice.
2. Lifecycle Approach to Process Validation
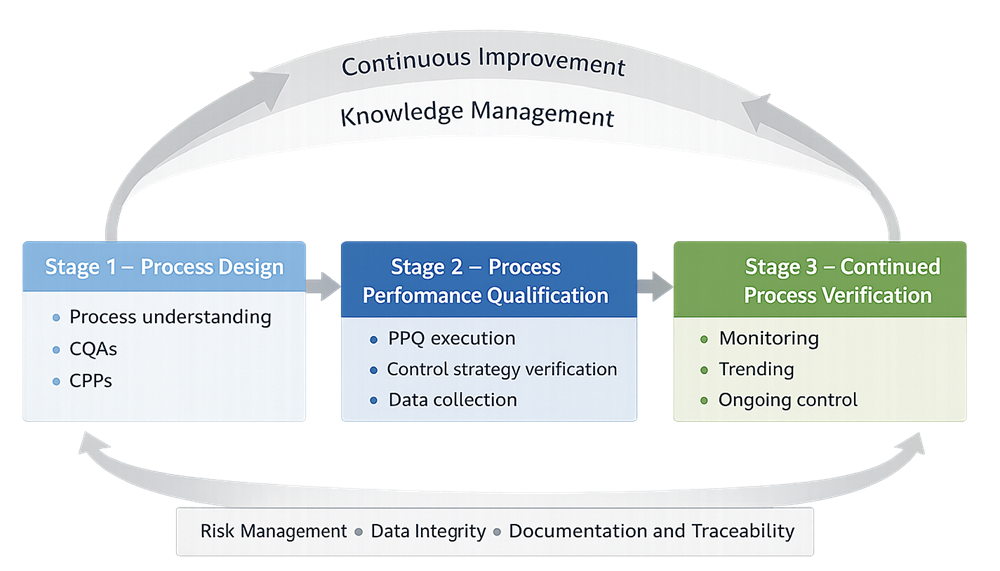
Process validation is not a single activity. It is a lifecycle approach consisting of three stages:
- Stage 1 — Process Design
- development and understanding of the manufacturing process
- identification of CQAs and CPPs
- Stage 2 — Process Performance Qualification
- demonstration that the process performs as intended under commercial conditions
- verification of control strategy
- Stage 3 — Continued Process Verification
- ongoing assurance that the process remains in a state of control
Each stage builds on the previous one and must be supported by data and documented evidence.
The following diagram illustrates the lifecycle approach to process validation, showing how process design, qualification, and continued verification are connected. It highlights the feedback loop that supports continuous improvement and the foundational systems that apply across all stages.
3. Process Understanding
Process validation is based on scientific understanding of how process inputs and parameters affect product quality. This includes:
- identification of CQAs
- identification of CPPs
- understanding of process variability
- knowledge of material and equipment impact
Process understanding is developed during process design and confirmed during PPQ.
4. Risk-Based Approach
Validation activities must be driven by risk to product quality. This requires:
- identification of critical variables
- evaluation of potential impact on CQAs
- prioritization of controls and testing
Risk assessment defines:
- what must be controlled
- how it must be controlled
- how it must be verified
5. Control Strategy
Control strategy translates process understanding and risk assessment into operational controls. It ensures that:
- CPPs are maintained within defined ranges
- CQAs consistently meet acceptance criteria
- variability is controlled
Control strategy must be implemented, verified during PPQ, and maintained during CPV.
6. Qualification and Verification
Process validation requires documented evidence that the process performs as intended. This includes:
- qualification of equipment and systems
- execution of PPQ protocols
- verification of process performance using defined acceptance criteria
Verification must demonstrate consistency, reproducibility, and control.
7. Data-Driven Decision Making
Validation conclusions must be based on objective data. This requires:
- representative sampling
- appropriate statistical methods
- evaluation of variability and trends
Decisions must be supported by data, not assumptions.
8. Continued Process Verification
Process validation does not end after PPQ. Ongoing monitoring is required to:
- confirm continued process control
- detect trends or shifts
- identify emerging risks
CPV ensures that the validated state is maintained throughout the product lifecycle.
9. Change Management
Changes to process, materials, equipment, or controls must be evaluated for impact on product quality. Requirements:
- assessment of impact on CQAs and CPPs
- application of risk-based evaluation
- implementation through formal change control
- verification where necessary
Uncontrolled change is a primary source of process failure.
10. Documentation and Traceability
All validation activities must be documented and traceable. This includes:
- linkage between process understanding, risk assessment, and controls
- documentation of execution and results
- traceability from raw data to conclusions
Documentation must demonstrate that validation is scientifically justified and consistently executed.
11. State of Control
The objective of process validation is to establish and maintain a state of control. A process is in a state of control when:
- CPPs operate within defined limits
- CQAs consistently meet specifications
- variability is understood and managed
- process performance is predictable
Maintaining a state of control requires continuous monitoring, evaluation, and improvement.

