Quantitative Method Capability
1. Purpose and Definition
Quantitative method capability defines the ability of an analytical method to measure residue levels accurately and precisely across a defined range. In cleaning validation, it ensures that reported values reflect the true amount of residue present and can be reliably compared to acceptance criteria.
This capability extends beyond detection and requires consistent numerical performance.
2. Role in Cleaning Validation
The analytical method must demonstrate that it can:
- quantify residues at and below acceptance limits
- produce consistent and reproducible results
- support decision-making based on measured values
- maintain performance across relevant concentration ranges
Detection alone is insufficient. Residues must be measurable with confidence.
3. Key Elements of Quantitative Capability
Quantitative capability is defined by the range over which the analytical response remains linear and reliable.
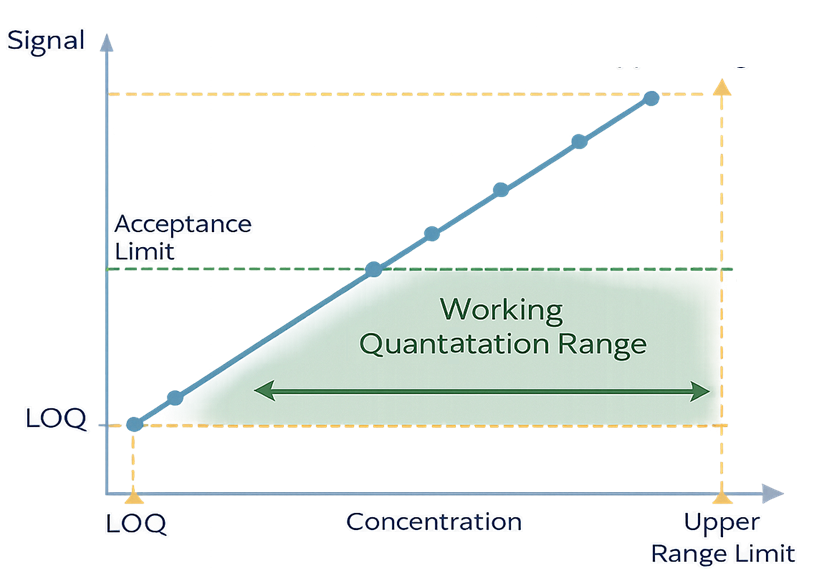
The method must demonstrate linear response across the range of interest. The lower boundary is defined by LOQ, while the upper boundary is defined by the validated range. Acceptance limits must fall within this range to ensure accurate quantitation. Limit of Quantitation (LOQ) defines the lowest concentration of analyte that can be quantitatively measured with acceptable accuracy and precision.
3.1 Accuracy
Accuracy reflects the closeness of measured values to the true value.
- evaluated using spiked samples
- must demonstrate minimal bias
- assessed across relevant concentration levels
3.2 Precision
Precision reflects the consistency of repeated measurements. Includes:
- repeatability
- intermediate precision
Results must show low variability under defined conditions.
Graph showing linear calibration curve with LOQ as lower boundary and acceptance limit within the validated quantitation range.
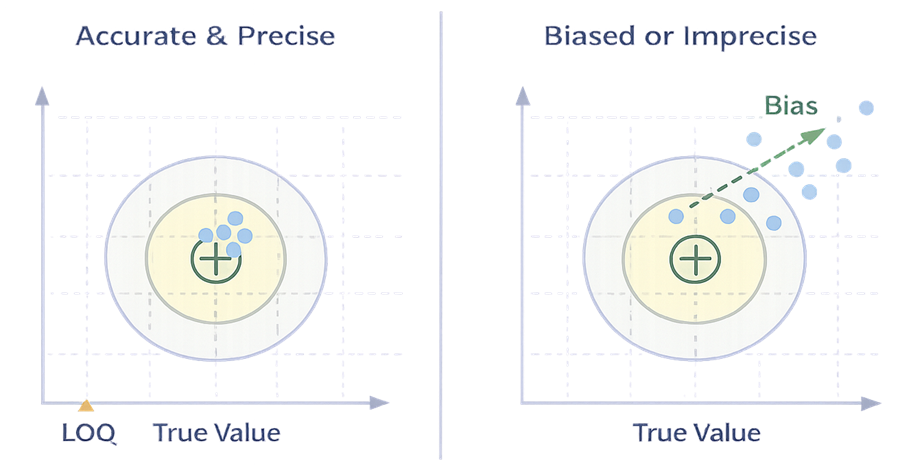
A method may detect residues at low levels but fail to measure them accurately or consistently. True quantitative capability requires both minimal bias and low variability, especially near the LOQ and acceptance limits.
3.3 Linearity
Linearity demonstrates proportional response between analyte concentration and measured signal.
- evaluated using calibration standards
- must cover the intended working range
- correlation must be justified
3.4 Range
The range defines the interval over which the method is accurate, precise, and linear. For cleaning validation, the range must include:
- LOQ
- expected residue levels
- acceptance limits
4. Relationship to Detection Limits
Quantitative capability is bounded by detection performance:
- Limit of Quantitation (LOQ) defines the lower limit of quantitation
- values below LOQ cannot be reliably measured
- method must perform adequately above LOQ
Detection limits alone do not confirm quantitative suitability.
5. Matrix and Sampling Effects
QQuantitative performance must be demonstrated under actual sample conditions, not only in standard solutions.
Analytical methods often perform well in clean calibration systems, but cleaning validation samples introduce additional complexity. The method must be evaluated using:
- swab extracts containing materials from sampling devices
- rinse solutions representing diluted and system-wide residues
- cleaning agents and excipients that may be present in the matrix
These components can alter analytical behavior in ways that are not predictable from calibration data alone.
Matrix effects may impact:
- signal response
suppression or enhancement of detector signal relative to true concentration - extraction efficiency
incomplete transfer of residue from swab or surface into solution - measurement accuracy
bias introduced by co-eluting or interfering substances
As a result, a method that appears accurate and precise in standard solutions may produce biased or inconsistent results in real samples.
Quantitative capability must therefore be confirmed using representative matrices that reflect actual cleaning validation conditions, ensuring that reported values remain accurate, precise, and reliable in practice.
6. Application of Recovery
Quantitative results are influenced by recovery efficiency.
- measured values may require correction
- recovery must be consistent and validated
- variability in recovery impacts quantitative reliability
Quantitative capability must consider the entire process, not only the analytical step.
7. Acceptance Criteria
The method must demonstrate:
- acceptable accuracy across range
- consistent precision
- linear response
- suitability at acceptance limit levels
Acceptance criteria must be predefined and justified.
8. Common Deficiencies
Typical issues include:
- acceptable LOQ but poor accuracy at low levels
- inadequate evaluation of precision
- non-linear response near acceptance limits
- failure to assess matrix effects
- inconsistent recovery application
These deficiencies compromise quantitative reliability.
9. Documentation Requirements
Quantitative capability must be documented. Required elements include:
- accuracy and precision data
- calibration and linearity data
- defined working range
- matrix evaluation results
- justification of method suitability
Documentation must support independent verification.
10. Lifecycle Considerations
Quantitative performance must be maintained over time.
Reassessment is required when:
- analytical methods are modified
- instruments are changed
- new matrices or products are introduced
Method capability must remain aligned with cleaning validation requirements.
11. Key Principle
Quantitative method capability ensures that measured values are reliable, not just detectable.
It confirms that analytical results can be used to demonstrate compliance with cleaning validation limits.

