Rinse Sampling
1. Purpose and Definition
Rinse sampling is an indirect sampling method used in cleaning validation to detect residues remaining within equipment systems after cleaning. It involves collecting a liquid rinse that has contacted internal surfaces and analyzing it for the presence of product residues.
The objective is to demonstrate that residues distributed across the system are below established acceptance limits derived from health-based criteria.
2. Regulatory and Scientific Basis
Rinse sampling is an accepted approach under 21 CFR 211.67 when scientifically justified.
It supports:
- evaluation of overall system cleanliness
- verification of clean-in-place (CIP) effectiveness
- assessment of inaccessible or complex internal surfaces
3. Sampling Principle
A suitable solvent is introduced into the equipment system after cleaning. The solvent contacts internal surfaces, mobilizes any remaining residues, and is then collected for analysis.
The result represents:
- cumulative residue from the entire contacted surface
- influenced by solvent effectiveness and flow coverage
- compared against predefined acceptance criteria
Rinse sampling provides system-wide data, not location-specific results. Rinse sampling relies on solvent contact with internal surfaces to mobilize and collect residues for analysis. The following diagram illustrates the process flow.
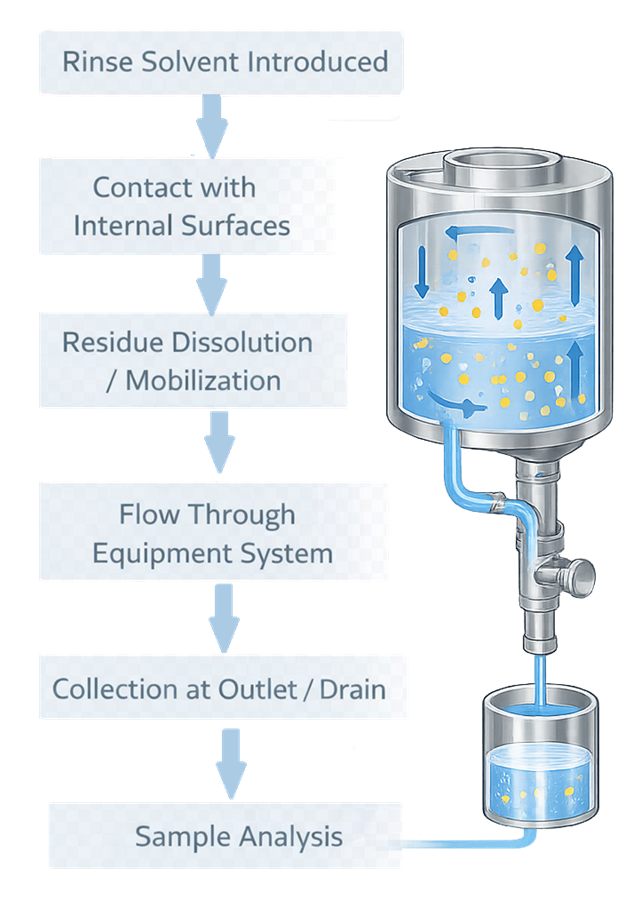
The solvent is introduced into the system and allowed to contact internal surfaces. Residues are dissolved or mobilized and transported through the system, where the rinse is collected at a defined outlet. The collected sample represents the cumulative residue from all contacted surfaces.
4. Selection of Rinse Conditions
Rinse conditions must be defined and justified.
Key parameters include:
- solvent selection
must effectively dissolve target residues - rinse volume
sufficient to contact all relevant surfaces - contact time
adequate for residue dissolution - flow dynamics
ensure coverage of internal surfaces
Conditions must represent or challenge actual cleaning processes.
5. Sample Collection
Rinse samples must be collected in a controlled and consistent manner.
Typical considerations:
- collection from defined outlet or drain point
- use of clean, inert containers
- avoidance of contamination during sampling
- documentation of volume and conditions
Sample integrity must be maintained from collection to analysis.
6. Recovery and Efficiency
Rinse sampling efficiency depends on the ability of the solvent to remove residues.
Influencing factors:
- residue solubility
- surface characteristics
- equipment geometry
- rinse flow and turbulence
Recovery studies may be required to demonstrate that the rinse method can adequately recover residues under defined conditions.
7. Analytical Considerations
Rinse samples must be compatible with analytical methods.
Requirements include:
- sufficient sensitivity to detect residues at acceptance limits
- absence of interference from solvent or matrix
- validated extraction and measurement
The analytical method must be capable of detecting diluted residues.
8. Advantages
Rinse sampling provides:
- coverage of entire system
- applicability to inaccessible areas
- suitability for CIP systems
- simplified sampling process compared to multiple swabs
9. Limitations
Rinse sampling has inherent limitations:
- dilution of residues may reduce detectability
- lack of location-specific information
- dependence on solvent effectiveness
- potential incomplete coverage of surfaces
It cannot replace swab sampling for worst-case location assessment.
10. Integration with Swab Sampling
Rinse and swab sampling are complementary:
- swab sampling → localized, worst-case surface evaluation
- rinse sampling → overall system cleanliness
A combined approach provides a more complete assessment of cleaning effectiveness.
Rinse and swab sampling provide different types of information and must be used together to ensure adequate coverage.

Swab sampling targets specific worst-case locations and provides localized, high-sensitivity data. Rinse sampling evaluates the entire system but represents a diluted and averaged measurement. Understanding this distinction is critical for proper interpretation of results.
11. Documentation Requirements
Rinse sampling must be fully documented.
Required elements:
- defined rinse conditions and justification
- solvent type and volume
- sampling location and method
- analytical method details
- raw data and calculated results
Traceability must support reconstruction of the sampling process.
12. Common Deficiencies
Typical issues include:
- inadequate solvent selection
- insufficient rinse volume or contact time
- lack of recovery justification
- poor control of sampling conditions
- inadequate analytical sensitivity
These deficiencies reduce confidence in results.
13. Lifecycle Considerations
Rinse sampling is used beyond initial validation:
- routine verification of CIP systems
- investigation of cleaning deviations
- periodic review and trending
Sampling strategy must evolve based on process knowledge and performance.
14. Key Principle
Rinse sampling provides system-wide evidence of cleanliness, but lacks specificity.
It must be used in conjunction with swab sampling to ensure comprehensive validation coverage.

