Shipping Validation
1. Purpose
This article defines the approach for validating shipping processes to ensure that pharmaceutical products are transported under controlled conditions without compromise to quality, safety, or efficacy. Shipping validation demonstrates that the combined packaging system and distribution process maintain product integrity throughout transit.
2. Regulatory Expectations
Shipping validation must demonstrate that distribution conditions do not adversely affect product quality in accordance with:
- 21 CFR Part 211 §211.150 for distribution practices
- ICH Q1A(R2) for environmental impact
- ICH Q9 for risk-based justification
- EU GMP Annex 1 where applicable
Validation must confirm that products remain within defined acceptance criteria during transport.
3. Scope of Shipping Validation
Shipping validation evaluates the full distribution system, including:
- packaged product configuration
- shipping containers and insulation systems
- transport modes such as ground, air, or sea
- handling conditions and logistics operations
The scope must represent actual or worst-case distribution scenarios.
4. Validation Approach
Shipping validation is performed using a combination of simulated and real-world studies. The diagram below illustrates how transport simulation and real shipment studies are combined to establish shipping validation.
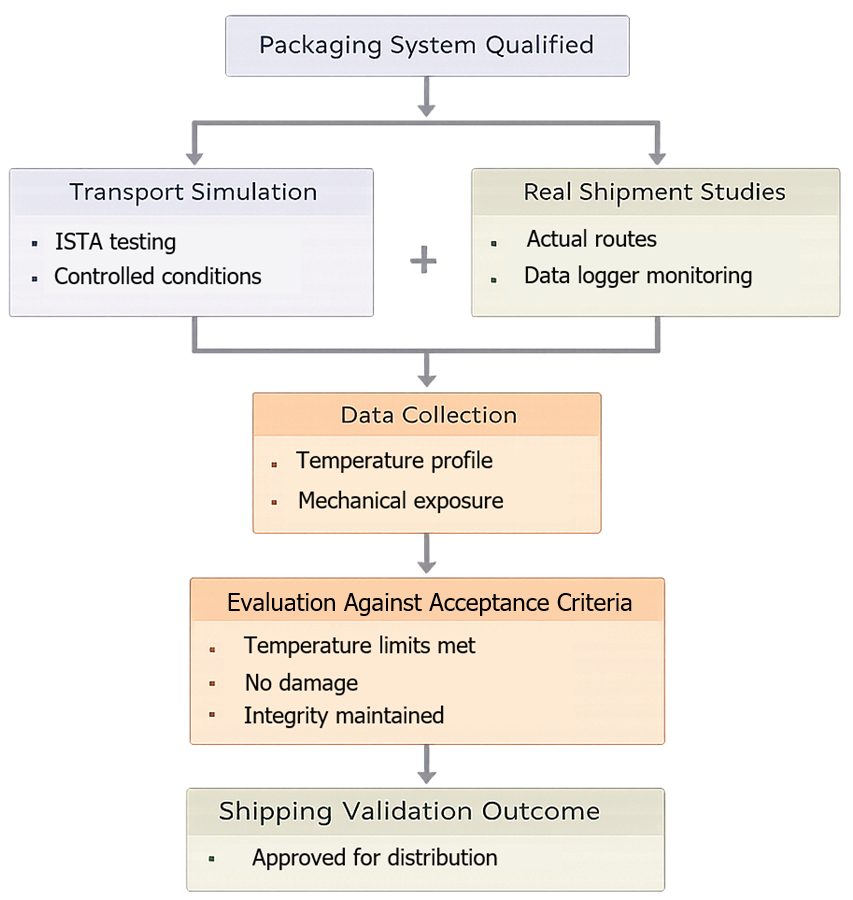
4.1 Transport Simulation
Laboratory-based testing using standardized protocols such as ISTA to simulate distribution hazards including vibration, shock, and compression.
4.2 Real Shipment Studies
Execution of shipments through defined distribution routes using actual logistics conditions. Data loggers are used to record environmental conditions during transit.
Both approaches are complementary. Simulation provides controlled repeatability, while real shipment studies confirm performance under actual conditions.
5. Environmental Control and Monitoring
For temperature-sensitive products, environmental conditions must be monitored throughout shipment.
Monitoring includes:
- temperature profiling using calibrated data loggers
- placement of sensors in worst-case locations
- recording of temperature excursions and duration
Data must demonstrate that required environmental conditions are maintained.
6. Worst-Case Conditions
Validation must include worst-case scenarios to ensure robustness of the shipping system.
Examples include:
- maximum shipment duration
- extreme ambient temperatures
- most challenging packaging configurations
- highest risk distribution routes
Worst-case justification must be documented and supported by data.
7. Acceptance Criteria
Acceptance criteria must be predefined and based on product requirements. Criteria typically include:
- maintenance of required temperature range where applicable
- absence of physical damage to packaging and product
- preservation of container closure integrity
- maintenance of labeling and identification
For temperature-controlled products, criteria must consider both excursion limits and cumulative impact where justified.
8. Data Analysis and Evaluation
Collected data must be evaluated to confirm compliance with acceptance criteria.
Evaluation includes:
- review of temperature profiles and excursion events
- comparison against defined limits
- assessment of impact on product quality
Data must support a clear conclusion regarding suitability of the shipping process.
9. Documentation and Traceability
All validation activities must be documented, including:
- defined shipping configurations and routes
- test methods and protocols
- monitoring setup and calibration
- results, deviations, and investigations
- final conclusions
Traceability must be maintained between packaging qualification, transport simulation, and shipping validation.
10. Outcome
The outcome of shipping validation is documented evidence that the packaging system and distribution process maintain product quality throughout transportation. This supports routine distribution and provides assurance that products reach the end user without compromise.

